| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://www.thejh.org |
Review
Volume 12, Number 4, August 2023, pages 145-160
Harnessing the Immune System: An Effective Way to Manage Diffuse Large B-Cell Lymphoma
Nathan Visweshwara, e, Juan Felipe Ricob, Robert Killeenc, Arumugam Manoharand
aDepartment of Hematology, University of South Florida, Tampa, FL, USA
bDepartment of Pediatrics, University of South Florida, Morsani College of Medicine, Tampa, FL, USA
cDepartment of Hematology, Moffitt Cancer Center, Tampa, FL, USA
dFaculty of Science, Medicine and Health, University of Wollongong, Wollongong, NSW, Australia
eCorresponding Author: Nathan Visweshwar, Department of Hematology, University of South Florida, Tampa, FL, USA
Manuscript submitted March 15, 2023, accepted May 1, 2023, published online August 31, 2023
Short title: Review of Immunotherapy for DLBCL
doi: https://doi.org/10.14740/jh1112
- Abstract
- Introduction
- Overview of Immunological Interventions in the Management of DLBCL
- Conclusion
- References
| Abstract | ▴Top |
Diffuse large B-cell lymphoma (DLBCL) is a heterogenous hematological disorder with malignant potential controlled by immunological characteristics of the tumor microenvironment. Rapid breakthrough in the molecular pathways has made immunological approaches the main anchor in the management of DLBCL, with or without chemotherapeutic agents. Rituximab was the first monoclonal antibody approved for the treatment of DLBCL. Following rituximab that transformed the therapeutic landscape, other novel immunological agents including chimeric antigen T-cell therapy have reshaped the management of relapsed/refractory DLBCL. However, resistance and refractory state remain a challenge in the management of DLBCL. For this literature review, we screened articles from Medline, Embase, Cochrane databases and the European/North American guidelines from March 2010 through October 2022 for DLBCL. Here we discuss immunological agents that will significantly affect future treatment of this aggressive type of lymphoma.
Keywords: Diffuse large B-cell lymphoma; Immunotherapy; Immunomodulatory therapy; Bispecific T-cell engager antibody; Antibody drug conjugates; Checkpoint inhibitors; Chimeric antigen receptor T-cell therapy
| Introduction | ▴Top |
Immunological interventions are used to target specific antigen of the malignant clone with the least lethal effect on the normal host tissue. However, the immune system also needs pro-effector environmental signals to unleash its lethal effect. Extensive efforts have been made to define antigenic targets differentiating “self” from “non-self”, but there appears to be no antigen unique to diffuse large B-cell lymphoma (DLBCL). With the monoclonal antibody rituximab, there was an improvement of nearly 15% 10-year overall survival (OS) when added to CHOP (cyclophosphamide, hydroxydaunorubicin, vincristine and prednisone) regimen [1]. After the initial breakthrough with rituximab, there has been no improvement in treatment of DLBCL as other novel CD20 antibodies including obinutuzumab or intensifying R-CHOP with 15-day regimen have not improved upon an every 21-day R-CHOP regimen, which remains the standard of care (SOC) for DLBCL treatment [2]. The long-term survival of DLBCL is 40-50%, and for the rest, there is an unmet need for improvement of quality of life and survival. This may be because of the substantial heterogeneity in the biology of DLBCL. Here, we discuss the role of immune-mediated treatment options, including immunomodulatory agents and cellular therapies in the management of DLBCL.
| Overview of Immunological Interventions in the Management of DLBCL | ▴Top |
Immunological interventions include immunomodulatory therapy, monoclonal antibodies, bispecific T-cell engager antibody, antibody drug conjugates (ADCs), checkpoint inhibitor therapy, and chimeric antigen receptor T cells (CARs) therapy (Fig. 1, Table 1 [3-25]).
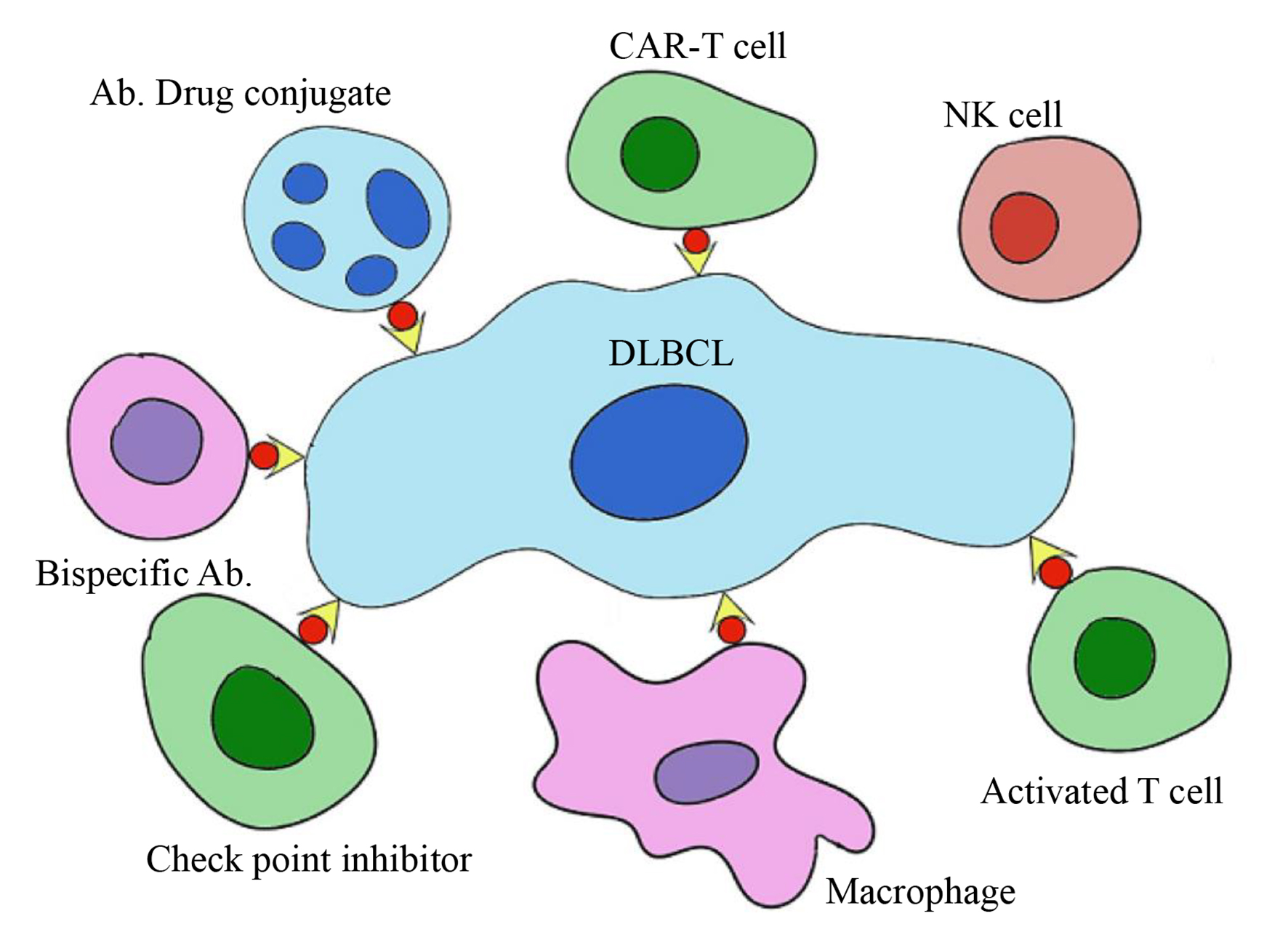 Click for large image | Figure 1. Immunological targets in DLBCL. DLBCL: diffuse large B-cell lymphoma. |
 Click to view | Table 1. Immune-Mediated Interventions in the Management of DLBCL |
Mechanism of action of the immunomodulatory agents
The immunomodulatory agents, lenalidomide and pomilidomide enhance the activity of B, T, NK, and dendritic cells and enhance antibody-dependent cellular cytotoxicity. Cereblon is identfied as a molecular target that may underlie the effects of lenalidomide on tumor cells, as well as on cells in the tumor microenvironment. Lenalidomide downregulates interferon regulatory factor 4 (the cell survival factor) and the gene SPIB that encodes SPI-B, a transcription factor which is upregulated in activated B cell (ABC)-DLBCL inducing cell death in ABC-DLBCL. Rituximab, the humanized chimeric anti-CD20 monoclonal antibody, induces killing of CD20+ cells via the direct effects of complement-mediated cytotoxicity and antibody-dependent cell-mediated cytotoxicity, and the indirect effects include structural changes, apoptosis, and sensitization of cancer cells to chemotherapy. Obinutuzumab, a type II anti-CD20, works primarily by inducing direct cell death by antibody-dependent cell-mediated cytotoxicity. Ofatumumab is a human monoclonal antibody against CD20 that mainly induces complement dependent cytotoxicity. Tafasitamab is a Fc-enhanced humanized CD19 antibody that induces NK cell-mediated lysis of B cells. Magrolimab, is a humanized, IgG4 isotype, CD47-blocking monoclonal antibody (CD47 antigen is a signal “do not eat me” for the macrophages), inducing phagocytosis of tumor cells by the blockade of CD47 and its ligand SIRPα. An antibody drug conjugate is a monoclonal antibody conjugated to the cytotoxic payload (usually a microtubule-disrupting agent) via a chemical linker that is directed toward a target antigen expressed on the lymphoma cell surface, reducing systemic exposure and toxicity. Bispecific antibodies, target two different antigens (CD19 or CD20 of the lymphoma cells and CD3 of the T-cell), to redirect an immune response to the tumor site, bypassing Fc-mediated or major histocompatibility complex (MHC)-restricted activation of effector cells. Bispecific antibody constructs are available off the shelf. Cancer cells escape immune recognition by exploiting the programmed cell death-1 (PD-1)/PD-1 ligands (PD-L1) immune checkpoint axis, and PD-1 blocking agents have changed the treatment landscape of classical HL and primary mediastinal B-cell lymphoma and the role of these agents in DLBCL is being evaluated. Chimeric antigen receptor redirected T cells (CARs) are autologous T cells genetically engineered to express CARs that can recognize a variety of tumor- associated antigens. The antigen-binding domain of B cell receptor is transfected to the intracellular domain of a CD3 TCR (CD3-zeta) via a viral vector. The CARs are engineered to target CD19/CD22 antigen of lymphoma cells and activate T cells, independently of MHC recognition.
Immunomodulatory agents
Immunomodulatory agents have become an important drug category in the treatment of DLBCL. These agents have a complex mechanism of action (Fig. 2).
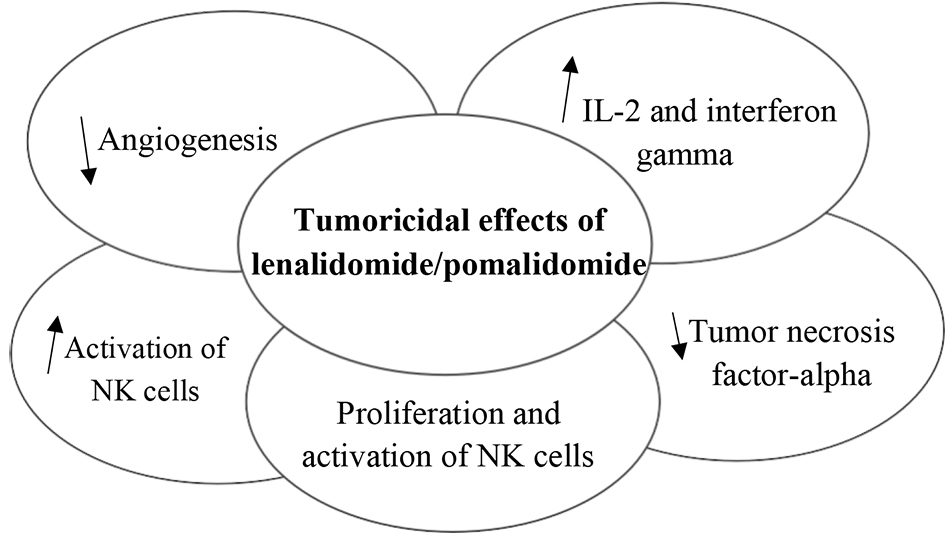 Click for large image | Figure 2. Effects of immunomodulatory agents. |
Lenalidomide
Lenalidomide is an orally active immunomodulatory drug that enhances the activity of B, T, natural killer (NK), and dendritic cells and enhances antibody-dependent cellular cytotoxicity. Cereblon, the molecular target is needed for the effects of lenalidomide on tumor cells, as well as on cells in the tumor microenvironment. Decreases in cereblon attenuate these effects and confer resistance to lenalidomide and antibody-dependent cellular cytotoxicity. These effects are secondary to cytokine production from T cells. Lenalidomide downregulates the cell survival factor, interferon regulatory factor 4, a transcription factor overexpressed in DLBCL providing its antitumor effects. Treatment with lenalidomide of activated B-cell DLBCL (ABC-DLBCL) resulted in downregulation of SPIB, the gene SPIB encodes SPI-B, a transcription factor upregulated in DLBCL. Treatment of ABC-DLBCL cells with lenalidomide induces rapid downregulation of SPIB mRNA transcripts and protein causing cell death [26]. Lenalidomide has been shown to produce synergistic effects in experimental models when evaluated in combination with rituximab, dexamethasone, bortezomib, and B-cell receptor signaling inhibitors, consistent with mechanisms complementary to these agents [27]. However, in ROBUST trial, patients with ABC-DLBCL who received lenalidomide plus standard R-CHOP21 (R2COP) versus placebo/R-CHOP21 for six cycles did not meet the primary end point of progression-free survival (PFS) (PFS for 2 years was 67% for R2COP and 64% for placebo/R2COP; hazard ratio (HR), 0.85; 95% confidence interval (CI): 0.63 - 1.14; P = 0.29) [28].
In an open-label, multicenter, phase 1b/2 study, 45 patients with relapsed/refractory (R/R) non-germinal DLBCL received ibrutinib plus lenalidomide and rituximab. The overall response rate (ORR) was 44% and complete response (CR) was 28% [29]. In a phase II study (L-MIND), 81 enrolled patients received tafasitamab + lenalidomide for R/R DLBCL. The ORR was 57.5% (n = 46/80), including CR in 40% of patients (n = 32/80) and partial response (PR) in 17.5% of patients (n = 14/80). The median duration of response (DoR) was 43.9 months (95% CI: 26.1 - not reached (NR), and NR in patients who achieved a CR (95% CI: 43.9 - NR); median PFS was 11.6 months (95% CI: 6.3 - 45.7), with median follow-up of 33.9 months; median OS was 33.5 months (95% CI: 18.3 - NR), with median follow-up of 42.7 months. There were no unexpected toxicities [3]. In a single-arm phase II trial of non-germinal center B-cell-like (GCB) DLBCL, 60 patients received rituximab, lenalidomide and ibrutinib. The ORR after two cycles was 86.2%, and the CR rate was 94.5%. The median PFS and OS were 91.3% and 96.6% at 2 years, respectively [30]. Lenalidomide was used in a phase Ib trial for refractory/recurrent central nervous system lymphoma with ibrutinib, and rituximab was evaluated for response with 13/15 (73%) showing a response: four CR, seven PR, and two stable disease (SD), one progression development (PD). Median PFS was 3.03 months [31]. Lenalidomide associated B-cell acute lymphoblastic leukemia (B-ALL) with high rates of TP53 mutation and hypodiploidy have been reported in myeloma but so far not reported in R/R DLBCL [32].
Pomalidomide (POM)
Pomilidomide is a second-generation immunomodulatory agent that induces cell cycle arrest and apoptosis of cells in neoplastic tissue. It has been used to treat relapsed and/or primary refractory human immunodeficiency virus (HIV)-associated DLBCL. In stage IV R/R DLBCL, 10 patients received pembrolizumab, an immune checkpoint inhibitor, with or without POM. The PFS was 4.1 months (95% CI: 1.3 - 12.4) and OS was 14.7 months (95% CI: 2.96 - NR) [4]. Thirty-three patients in a phase Ia/Ib study enrolled with R/R DLBCL (n = 8), chronic lymphatic leukemia/small lymphocytic lymphoma (CLL/SLL) (n = 5), Richter’s transformation (RT) (n = 6), follicular lymphoma (FL) (n = 5), mantle cell lymphoma (MCL) (n = 4), marginal zone lymphoma/lymphoplasmacytic lymphoma (MZL/LPL) (n = 3), and Hodgkin’s lymphoma (HL) (n = 2) received an optimized oral triplet combination of a novel BTK inhibitor DTRMWXHS-12 (DTRM-12) with everolimus and POM. About 48% of all patients had a ≥ 50% reduction in sum of the products of lymph node diameter [33].
Monoclonal antibodies
As part of the induction regimen for DLBCL, monoclonal antibodies provide additional benefit (Fig. 3).
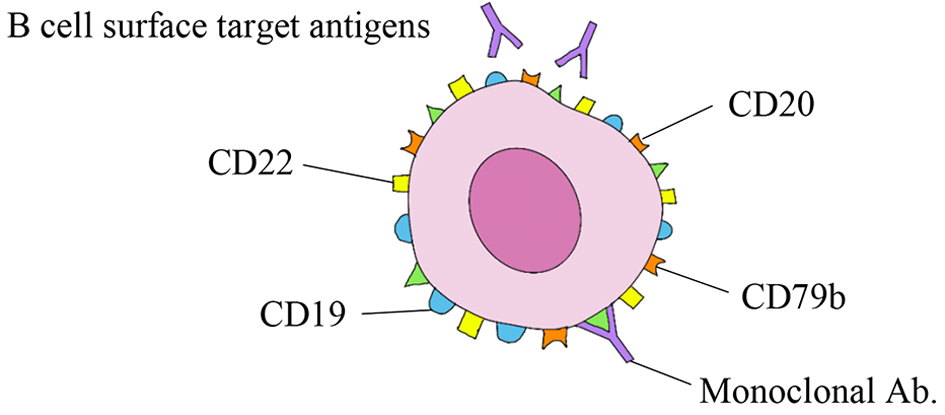 Click for large image | Figure 3. Monoclonal antibodies. |
Rituximab
Rituximab is a chimeric anti-CD20 monoclonal antibody that decreases B-cell levels in peripheral lymph nodes and bone marrow (as 95% depletion of B cells in peripheral lymph nodes and bone marrow requiring 60 to 90 days to attain normal levels and improves survival in DLBCL [34]. The addition of rituximab to the CHOP regimen increases the CR rate and prolongs OS in elderly patients with DLBCL, without a clinically significant increase in toxicity [5]. The ORR of rituximab when added to CHOP was 94% [35]. Rituximab to CHOP chemotherapy has resulted in improvement in outcome for DLBCL irrespective of age with the event-free survival (EFS), PFS and OS not different between six versus eight cycles of R-CHOP-14 for DLBCL and it added to the toxicity [36, 37]. Rituximab maintenance therapy improves survival which was not statistically significant in patients with DLBCL [38].
Obinutuzumab
Obinutuzumab is a type II anti-CD20 monoclonal antibody that induces direct cell death and has antibody-dependent cellular cytotoxicity than rituximab that is used in combination with other investigational agents in first-line FL and DLBCL. A flat dose of 1,000 mg for obinutuzumab rather than conventional body surface area-based dosing is advised [6]. A trial with 25 DLBCL and 15 MCL R/R showed response to obinutuzumab monotherapy. Among 40 patients, 21 patients were in the 400/400 mg treatment arm (DLBCL, n = 10; MCL, n = 11) and 19 patients were in the 1,600/800 mg arm (DLBCL, n = 15; MCL, n = 4). The ORR was 37% in the 1,600/800 mg arm and 24% in the 400/400 mg study arm [39]. There are various open-label, multicenter phase Ib/II studies with other investigational agents including atezolizumab plus obinutuzumab and CHOP or bendamustine in first-line FL and DLBCL [40]. Obinutuzumab-CHOP (G-CHOP) did not significantly improve PFS compared with R-CHOP in previously untreated patients with DLBCL and serious adverse events (43% versus 38%) were also more common in the G-CHOP than in the R-CHOP arm [41].
Ofatumumab
Ofatumumab is a human monoclonal antibody against CD20 that induces complement dependent cytotoxicity. In a phase II trial of elderly patients with newly diagnosed DLBCL the ORR was 90.5% and the CRR was 33.3%. Median PFS and OS were 8.6 and 12.0 months, respectively [42]. A phase II study of 85 patients with R/R DLBCL patients eligible/transplant ineligible (TI) for autologous stem cell transplantation (ASCT) received ofatumumab 300 mg followed by seven weekly IV infusions of 1,000 mg. The ORR was 13% for the TI group (seven PRs) and 8% for the relapse/progression after transplantation (PT) group (two CRs) [12]. Patients with R/R CD20+ DLBCL were assigned to three cycles of rituximab-dexamethasone, cytarabine, cisplatin (DHAP) or ofatumumab-DHAP. Either ofatumumab 1,000 mg or rituximab 375 mg/m2 was administered for a total of four infusions (days 1 and 8 of cycle 1; day 1 of cycles 2 and 3 of DHAP). PFS at 2 years was 24% versus 26% and there was no difference in efficacy between these two groups [43].
Targeting NK cells with Fc-enhanced CD19 antibody (tafasitamab)
The CD19 antigen is expressed on DLBCL cells, and the Fc-enhanced, humanized, anti-CD19 monoclonal antibody tafasitamab induces NK cell-mediated lysis of B cells in lymphoma and leukemia. This agent showed promising clinical activity in patients with R/R DLBCL [44]. In a phase II study tafasitamab showed clinical activity in patients with R/R DLBCL with responses in five out of nine patients lasting for more than 12 months, including in patients with rituximab refractory lymphoma [10]. In a phase II study, tafasitamab with lenalidomide was well tolerated and resulted in a high proportion of patients with R/R DLBCL ineligible for ASCT having a CR, and might represent a new therapeutic option in this setting [3].
Targeting macrophages with anti-CD47 antibody (magrolimab)
The M1 macrophages are immunosuppressive and aid tumor growth (Fig. 4). The antigen CD47 is an antiphagocytic signal that is overexpressed on circulating hematopoietic stem cells, lymphoma and leukemia cells to avoid phagocytosis [45]. Magrolimab, a humanized, IgG4 isotype, CD47-blocking monoclonal antibody, induces phagocytosis of tumor cells by the blockade of CD47 and its ligand SIRPα [46]. The anti-CD47-induced phagocytosis by M1 was more prominent than that for M2 [47]. The combination of magrolimab and rituximab demonstrated safety and efficacy in a cohort of patients with R/R DLBCL. Among patients with DLBCL, the CD47 expression is consistently high with the objective response and CR rates of 40% and 33%, respectively with magrolimab. At a median follow-up of 6.2 months among patients with DLBCL, 91% of the responses were ongoing [21].
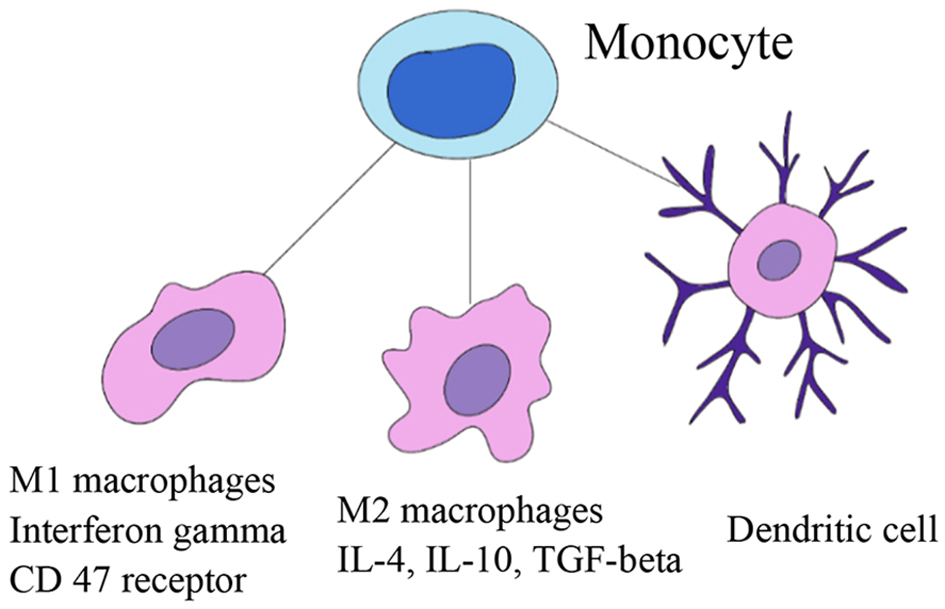 Click for large image | Figure 4. Targeting macrophages with anti-CD47 antibody (magrolimab). |
Bispecific antibodies
Bispecific antibodies, consisting of two different antigens, are used in treatment of R/R DLBCL to redirect an immune response to the tumor site, bypassing Fc-mediated or MHC-restricted activation of effector cells. The initial clinical studies were disappointing due to low efficacy and severe adverse effects in DLBCL. The retargeting is achieved by simultaneous binding of the bispecific molecule to a tumor antigen on the target cell (CD19) and a trigger molecule on the effector cell (CD3), leading to tumor cell destruction [48]. With recombinant antibody technology the current bispecific antibodies consist of several smaller formats where the antibody is essentially reduced to the domains involved directly in the antigen binding [49]. Thus, the redirection of an immune response to the tumor site, bypassing Fc-mediated or MHC-restricted activation of effector cells, is achieved. In this context, CD3 has become widely accepted as the appropriate trigger molecule [48].
Blinatumomab
Blinatumomab, a bispecific T cell engaging antibody (CD19 and CD3), directed against the B cell differentiation antigen showed remarkable single-agent activity in patients with R/R DLBCL and R/R B-cell precursor ALL [11]. Cytokine release syndrome and neurological side effects were dose-limiting. Adverse effects were manageable and transient in nature. Based on the results of an international phase II trial, blinatumomab received FDA approval for the treatment of R/R B-precursor ALL in December 2014 [11]. In a phase III trial of adults with heavily pretreated B-cell precursor ALL, of the 37 patients with minimal residual disease (MRD) positivity, 27 patients (73%) achieved MRD-negative remission with blinatumomab. With a median follow-up of 31 months (range, 5 - 70 months), the 3-year OS rate was 67% (95% CI: 46-81) [50]. Consolidation with blinatumomab in patients with newly diagnosed, high-risk DLBCL who did not progress under R-CHOP was better tolerated than in previous studies in patients with high-risk lymphoma [51]. Richter’s syndrome is an aggressive transformation of CLL, clonally related to DLBCL. In a phase II study of blinatumomab four out of nine patients had reduction in nodal disease, including one CR lasting > 1 year [52].
Mosunetuzumab
Mosunetuzumab, a fully humanized immunoglobulin G1 (IgG1) bispecific antibody targeting both CD3 and CD20, induces complete remissions in poor prognosis DLBCL, including those who are resistant to or relapse after CARs therapy. In a phase I/II, open-label, multicenter study, 19 patients with DLBCL who were unable to tolerate full-dose chemotherapy received single-agent mosunetuzumab, the ORR was 58% (11/19) and CR rate was 42% (8/19) [53]. A total of 43 patients with R/R DLBCL and newly diagnosed DLBCL received mosunetuzumab-CHOP (M-CHOP), seven patients with R/R DLBCL, and 36 patients with newly diagnosed DLBCL. In R/R non-Hodgkin lymphoma (NHL) treated with M-CHOP (n = 7), the ORR was 86%, with 71% of patients achieving a CR [54]. In a phase I/Ib, dose escalation study of mosunetuzumab in R/R B-cell NHL, the ORR and CR rates were 43.8% (7/16) and 25.0% (4/16, 2 DLBCL and 2 FL), respectively [13].
Plamotamab
Plamotamab is a humanized bispecific antibody that binds both CD20 and CD3 to recruit cytotoxic T cells to kill CD20 expressing malignant cells. In this phase I study of 80 patients with R/R NHL, there were 23 objective responses (43.4% OR) [55]. A randomized, multicenter, open-label, two-part study is in progress comparing the safety and efficacy of plamotamab, tafasitamab, lenolidamide vs. tafasitamab, lenolidamide in adult subjects with DLBCL who have relapsed or are refractory to ≥ 1 prior line of therapy and are ineligible for or refuse autologous stem cell therapy [15].
Epcoritamab
Epcoritamab is a novel bispecific IgG1 antibody redirecting T-cells toward CD20+ tumor cells, expressed on all mature B-cells, but absent on hematopoietic stem cells, pro-B-cells, and plasma cells. In a phase I trial adults with previously untreated DLBCL and an R-IPI score ≥ 3 received flat-dose epcoritamab in combination with standard R-CHOP for six cycles followed by epcoritamab monotherapy. Of the nine patients (four patients with epcoritamab 24 mg; five patients with epcoritamab 48 mg) with median age of 66 (range, 56 - 78), four patients had response assessment, with three patients achieving complete metabolic response. No grade 3 CRS events or cases of febrile neutropenia were reported [56]. In a phase I/II trial including 73 patients with R/R DLBCL, 68 patients received escalating full doses (0.0128 - 60 mg) of subcutaneous epcoritamab, the ORR in patients with R/R DLBCL was 68% (95% CI: 45-86), with 45% achieving a CR at full doses of 12 - 60 mg [16].
Odronextamab
Odronextamab is a hinge-stabilized, fully human IgG4-based CD3 × CD20 bispecific antibody that has demonstrated encouraging safety, and efficacy in patients with R/R DLBCL. In a phase I study, 127 patients with R/R DLBCL were treated at doses ranging from 0.03 to 320 mg. The study included 71 patients with DLBCL, who had not received prior CARs therapy, treated at doses ≥ 80 mg (n = 10); ORR and CR rates were 60%; median observed DoR was 10.3 months (range, 2.9 - 18.6+), with four of six CRs ongoing at last tumor assessment. The median duration of clinical remission was 9.5 months (range, 2.9 - 18.6+) and follow-up is ongoing. In DLBCL patients who were refractory to prior CARs therapy, treated at doses ≥ 80 mg (n = 21), ORR was 33.3%, and CR rate was 23.8%; median observed DoR was 2.8 months (range, 0+ to 18.9) [57]. Odronextamab monotherapy showed a manageable safety profile and encouraging preliminary activity, including durable responses in heavily pretreated patients with R/R DLBCL. In a phase I study of 145 patients with R/R DLBCL with single agent odronextamab without previous CARs therapy who received doses of 80 mg or higher, the objective response rate was 53% (8/15) and all responses were CRs. In patients with R/R DLBCL who had previous CARs therapy and received doses of 80 mg or higher, the objective response rate was 33% (10/30) and CR rate was 27% (8/30) [17].
Glofitamab
Glofitamab is a T-cell-engaging bispecific antibody possessing a novel 2:1 structure with bivalency for CD20 on B cells and monovalency for CD3 on T cells. In a phase I study evaluating glofitamab in 127 patients with RR/ DLBCL, the ORR was 53.8% (CR, 36.8%) among all doses and 65.7% (CR, 57.1%) in those dosed at the recommended phase II dose. Of 63 patients with CR, 53 (84.1%) patients have ongoing CR with a maximum of 27.4 months observation [58]. In a phase II expansion study, 107 patients with R/R DLBCL (35% had received prior CAR-T cell therapy) were given single agent glofitamab. After a median follow-up of 9 months (0.1 - 16), ORR and CR rates by IRC were 50.0% and 35.2%, respectively. CR rates were consistent in patients with and without prior CAR-T cell therapy (32% versus 37%). The median time to CR was 42 days (95% CI: 41 - 48). The majority of CRs (33/38; 87%) were ongoing at data cut. An estimated 84% of complete responders and 61% of responders remained in response at 9 months [14].
Antibody drug conjugate (ADC)
An ADC is a monoclonal antibody attached to a cytotoxic agent via a chemical linker (Fig. 5). The ADCs are used to treat R/R DLBCL by selectively ablating tumor cells and ameliorating the therapeutic index of cytotoxic drugs. The target, the cytotoxic agent, and the way the agent is attached to the antibody determine the pharmacokinetics, clinical effect, and tolerability [59]. ADCs are a promising class of drugs with the potential to specifically target tumor cells and ameliorate the therapeutic index of cytotoxic drugs. In recent years, three ADCs, brentuximab vedotin (BV), polatuzumab vedotin, and loncastuximab tesirine, are approved for treatment of DLBCL. The ADCs can also diffuse into adjacent cells even if the cells are target-negative, resulting in cell death [60]. There was no statistical correlation between response and level of antigen expression; however, all responding patients had quantifiable CD30 by computer-assisted assessment of immunohistochemistry. It was thought that the bystander killing mechanism of an ADC involved surface antigen targeting, internalization, intracellular linker cleavage, drug release, and diffusion of drug away from the targeted cell. However, other mechanisms may be involved [61]. ADCs are cleaved extracellularly by other mechanisms than direct antigen-internalization by cathepsin. Increased levels of cathepsin B secretion are seen in tumor cells and high expression of cathepsin is also seen in macrophages and stromal fibroblasts [62, 63].
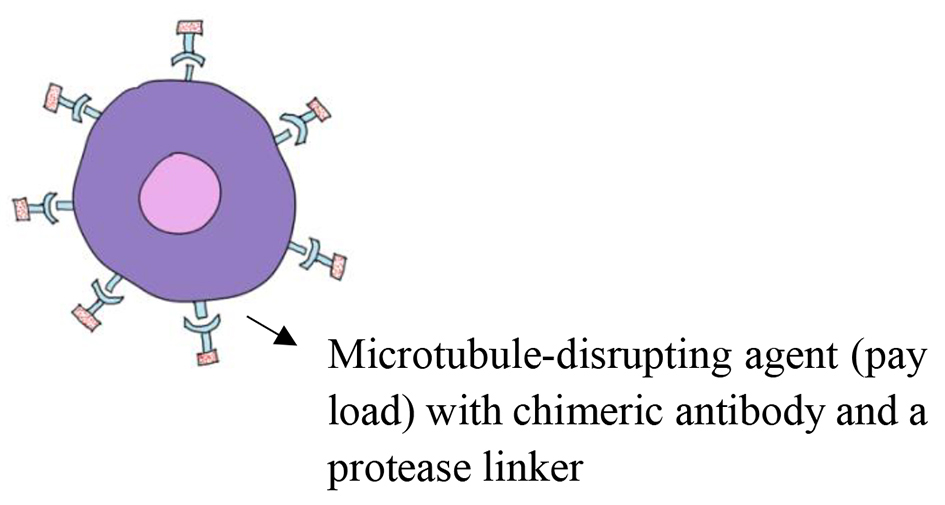 Click for large image | Figure 5. Antibody-drug conjugate. |
Brentuximab Vedotin (BV)
Brentuximab Vedotin is a CD30-directed monoclonal antibody conjugated to the potent inhibitor of microtubule polymerization monomethyl auristatin E (MMAE) - effective in targeting and killing CD30-expressing lymphoma for patients with relapsed HL and relapsed systemic anaplastic large-cell lymphoma [19]. In a phase II study of R/R high-CD30 expressing high grade lymphoma (anaplastic large cell lymphoma), BV produced disease control rate of 48.5% (16/33) including six CR and six PR; six patients (four CR, two PR) maintained over 16 completed cycles. Response to BV and survival were not associated with CD30 expression levels. Over a median follow-up of 29.2 months, the median PFS and OS rates were 1.9 months and 6.1 months, respectively [64]. A phase II clinical trial evaluated the efficacy and safety of BV in patients with R/R CD30+ primary mediastinal B cell lymphoma. The ORR was 13.3% (2/15), two patients achieved PR, one patient had SD, and the remaining 12 patients had PD. It was decided that BV had very low antitumor activity in this setting [65]. A phase II study evaluated BV monotherapy in 52 patients with CD30-expressing DLBCL. 16 of 52 patients with undetectable CD30 had an objective response, and the median OS was 7.5 months. Correlative analyses of CD30-undetected and CD30-expressing DLBCL combined demonstrated that ≥ 1% CD30 expression by conventional immunohistochemistry (cIHC) resulted in a trend toward a higher response rate and significantly longer median PFS and OS [66]. In a phase II study of R/R DLBCL with variable CD30 expression, 49 patients with DLBCL were treated with BV, and the objective response rate was 44% for DLBCL, including eight (17%) CRs with a median DoR of 16.6 months thus far (range, 2.7 - 22.7+ months) [19]. In a phase 1 trial in which 37 patients with R/R DLBCL were treated with BV, lenalidomide and rituximab, the ORR was 56.7%. The PFS and median OS were 11.2 months and 14.3 months, respectively and results were similar in the CD30+ and CD30 < 1% [67, 68]. In a phase II study, 30 patients with primary mediastinal large B-cell lymphoma (PMBCL) were treated with checkpoint inhibitor nivolumab and BV. At a median follow-up of 11.1 months, the ORR was 70% (51% to 85%), with a 43% complete metabolic response [69].
Inotuzumab ozogamicin (InO)
Inotuzumab Ozogamicn is an anti-CD22 monoclonal antibody-calicheamicin conjugate that binds to CD22-expressing tumor cells with cytotoxic calicheamicin derivative inducing double-strand DNA breakage and subsequent cell death. It was shown to be effective in treating aggressive histology lymphoma and DCBCL [18]. In a phase I study of InO, rituximab, cyclophosphamide, vincristine, and prednisone (R-CVP), in patients with R/R CD22+ B-cell NHL, the ORR was 57% for those with DLBCL [70]. In a randomized trial of InO plus rituximab (166 patients) versus chemotherapy plus rituximab (172 patients) for R/R DLBCL, the median OS and PFS with R-InO were 9.5 (95% CI: 7.0 - 14.5) and 3.7 (2.9 - 5.0) months, respectively [71]. In a phase 1 study, InO combined with rituximab, gemcitabine, dexamethasone, and cisplatin (R-GDP) in 21 patients with R/R CD22+ DLBCL B-cell NHL, the overall (objective) response rate was 33% [72]. In a phase I trial of relapsed R/R DLBCL treated with InO, the objective response rate at the end of treatment was 15% for all patients with DLBCL, and the median PFS was 49 days [73]. In a phase II study of InO and rituximab, followed by ASCT in patients with R/R DLBCL, 18 patients underwent HDT-ASCT; 2-year PFS for these patients was 61.1% [74].
Polatuzumab vedotin (Pola)
Polatuzumab vedotin is an ADC that delivers MMAE, a microtubule inhibitor, approved for the treatment of R/R DLBCL transplant ineligible patients [75]. Polatuzumab vedotin plus bendamustine and rituximab (pola + BR) received regulatory approvals for R/R DLBCL based on primary results from the randomized arms of the study [76]. In a phase 1b/2 study, 106 patients with R/R DLBCL were treated with polatuzumab, bendamustine and rituximab. The objective response rate was 41.5% and the CR rate was 38.7%. The PFS and OS were 6.6 and 12.5 months, respectively [76]. In a multicenter, open-label study of polatuzumab combined with bendamustine and rituximab or _obinatzumab_ in patients with R/R DLBCL the ORR was 68% versus 50%, CR of 36% versus 33% and PR of 32% versus 17% [77]. Polatuzumab vedotin plus bendamustine and rituximab was effective for patients with R/R DLBCL, with a median OS of 8.7 months and PFS of 3.8 months [78]. In 14 Israeli centers of the 47 patients (31 patients with de-novo DLBCL and 16 patients with transformed lymphoma) treated with polatuzumab-based regimen, the ORR was 61% (29), including 40% (19) CR and 21% (10) PR. The median OS and PFS were 8.3 and 5.6 months, respectively [79]. In a study of 51 patients with R/R DLBCL, treated with polatuzumab, bendamustine and rituximab to bridge to CARs therapy, 6-month PFS and OS were 27.7% and 49.6%, respectively. In the CARs cohort, OS at 6 months was 77.9% [80]. Polatuzumab-based therapy at multiple centers for its efficacy analyzed in USA in patients with R/R DLBCL revealed that the response rate was 50%, including 24% CR. Median DoR was 5.1 months, PFS was 2.0 months, and OS was 5.3 months [81]. In a multi-center retrospective study of polatuzumab for patients with DLBCL who relapsed after SOC CARs therapy, a response was achieved in 45% patients, including CR in 14% patients and PR in 30% with a median OS of 16 weeks [82]. In the United Kingdom, patients with DLBCL (133 patients) ineligible for stem cell transplantation who received polatuzumab vedotin with bendamustine and rituximab, had an ORR of 57.0% (32.8% CR) [20].
Loncastuximab tesirine
Loncastuximab tesirine is an ADC approved for the treatment of B-cell lymphoma. Loncastuximab tesirine consists of a pyrrolobenzodiazepine DNA-alkylating warhead covalently attached via a cleavable linker to an anti-CD19 antibody that binds to B cells [83]. In a study, 183 patients received loncastuximab tesirine, with 3 + 3 dose escalation at 15 to 200 g/kg and dose expansion at 120 and 150 g/kg. The ORR was 45.6%, including 26.7% CRs. The ORR in patients with DLBCL, MCL, and FL were 42.3%, 46.7%, and 78.6%, respectively. Median DoR in all patients was 5.4 months and not reached in patients with DLBCL (doses ≥ 120 µg/kg) who achieved a CR [84]. In response assessment low albumin, bulky disease, and mild/moderate hepatic impairment had a negative effect on OS [85]. In a phase 2 study, loncastuximab tesirine plus ibrutinib improved the survival of patients with DLBCL (35, 22, and 13 patients with DLBCL overall, non- GCB DLBCL, and GCB DLBCL), the ORR (CR + PR) was 57.1% (20/35; 95% CI: 39.4-73.7) in the overall DLBCL cohort, and 45.5% (10/22; 95% CI: 24.4-67.8) and 76.9% (10/13; 95% CI: 46.2-95.0) in the non-GCB DLBCL and GCB DLBCL cohorts, respectively [9].
Checkpoint inhibitor therapy
Cancer cells escape immune recognition by exploiting the programmed cell death-1 (PD-1)/PD-1 ligands (PD-L1) immune checkpoint axis, and PD-1 blocking agents have changed the treatment landscape of classical HL and primary mediastinal B-cell lymphoma and the role of these agents in DLBCL is being evaluated [86]. In the treatment of primary mediastinal B-cell lymphoma with genetic aberrations at 9p24 and upregulation of PD-L1, FDA approved PD-1 blocking agents.. However, unlike HL which is characterized by universal genetic alterations in 9p24.1, resulting in constitutive expression of PD-1 ligands, non-HL does not display a high frequency of 9p24.1 alterations which upregulates PD-L1 ligand, hence most of the patients with DLBCL do not respond to PD-1 blockade [87].
Pembrolizumab
Pembrolizumab, a selective humanized monoclonal antibody blocks the interaction between PD-1 expressed by tumor-associated T-cells and its ligand PD-L1 present on tumor and stromal cells. Therapy with combined pembrolizumab with R-CHOP in 30 previously untreated DLBCL patients at median follow-up of 36 months, the estimated PFS is 83% and OS is 86% [88]. In a phase I/II study, pembrolizumab was given every three weeks to eight patients with progressive DLBCL after anti-CD19 directed therapy. The ORR was 25%, with one patient having continued CR at 280 days [89]. Pembrolizumab was found to be very useful in the management of R/R PMBCL. RT of chronic lymphatic leukemia to DLBCL may be either de novo or a therapy-related process, with fludarabine possibly playing a role in reactivating Epstein-Barr virus [90]. In a phase 2 study, pembrolizumab, at a dose of 200 mg every 3 weeks, was given in relapsed and transformed CLL (16 relapsed and nine RT). Objective responses were observed in four out of nine RT patients (44%) and in zero out of 16 CLL patients (0%). All responses were observed in RT patients who had progression after prior therapy with ibrutinib. After a median follow-up of 11 months, the median OS in the RT cohort was 10.7 months but was not reached in RT patients who progressed after prior ibrutinib [91].
Nivolumab
In a phase I, open-label, dose-escalation, cohort-expansion study of 11 patients with R/R DLBCL, nivolumab (humanized monoclonal antibody against PD1) achieved an objective response rate of 36% and the DoR ranged from 6.0 to 81.6 weeks [23]. In a phase II study of 121 patients, nivolumab achieved an objective response rate of 10% and a DoR of 11 months [92]. In a trial with combined checkpoint inhibition with ipilimumab and nivolumab as consolidation following ASCT of seven patients with R/R DLBCL (43% of which were not in CR at time of ASCT), patients had a PFS at 18 months of 83% with checkpoint inhibitor therapy, while the historical reported PFS at 18 months with ASCT alone is 50% [93].
Avelumab
Avelumab is a monoclonal antibody targeting PD-1 ligand. In a phase II study, 28 patients with R/R DLBCL were given avelumab/_zacyti induction for two cycles q2-weekly (Av 10 mg/kg IV + R 375 mg/m2 IV), followed by R-CHOP21 for six cycles then avelumab 10 mg/kg for six cycles q2-weekly if in complete metabolic response post R-CHOP. ORR post R-CHOP was 89% (all CR). With a median follow-up of 16 months, 1-year PFS was 76% and OS 89% [94]. In a multicenter, randomized, open-label, parallel-arm Ib study 29 adult patients with R/R DLBCL were randomized 1:1:1 to receive avelumab in combination with utomilumab (an immunoglobulin G2 4-1BB agonist) and rituximab (arm A), avelumab in combination with utomilumab and _zacytidine (arm B), or avelumab in combination with bendamustine and rituximab (arm C). As the antitumor activity in arms A and B was minimal (one PR in arm A, no responses in arm B and three CRs in arm C) the study was discontinued [24].
Durvalumab
In a phase II open-label trial, 46 patients received durvalumab (a programmed death-ligand 1 inhibitor), combined with R-CHOP or lenalidomide + R-CHOP (R2-CHOP) - 43 received R-CHOP and three R2-CHOP. In this study 54.1% patients receiving durvalumab + R-CHOP achieved CR, 18.9% PR; 67.6% (25 patients) continued to consolidation and were progression-free at 12 months [95]. Durvalumab was evaluated with either tremelimumab (an immune checkpoint blocker) or danvatirsen (a 16-nucleotide antisense oligonucleotide) in a phase Ib dose escalation and dose expansion study. In this study, 32 patients were enrolled with overall objective response rate of 6.3%, with two PRs. Median time to response was 11.0 weeks with median PFS of 7.4 weeks and median OS of 28.0 weeks [25].
Chimeric antigen receptor redirected T cells (CARs)
Chimeric antigen receptor redirected T cells are autologous T cells genetically engineered to express CARs that can recognize a variety of tumor-associated antigens (Fig. 6). They have shown remarkable success in treating patients with R/R DLBCL who have failed to respond to standard chemotherapy or relapsed after ASCT [96]. The antigen-binding domain of B cell receptor is transfected to the intracellular domain of a CD3 TCR (CD3-zeta) via a viral vector (Fig. 6). The CARs are engineered to target a specific cell surface antigen and activate T cells independently of MHC recognition (Fig. 6). Various modifications can enhance CAR effector function, such as co-expression of intracellular costimulatory domains such as CD28 or 4-1BB (CD137) [97]. They have shown remarkable success in treating patients with R/R DLBCL who have failed to respond to standard chemotherapy or relapsed after ASCT. The primary side effects of CARs include cytokine release syndrome with fever, hypotension, altered mental status, and seizures, with some patients requiring intensive care [98]. In DLBCL unresponsiveness to standard chemotherapy and relapse after ASCT are indicators of poor prognosis and the CARs are emerging as a salvage treatment for these patients [99]. Anti-CD19 CARs have activity against chemotherapy-refractory lymphoma with durable complete remissions lasting > 2 years in about 50% of patients with R/R DLBCL, whereas historically the median OS of R/R DLBCL is between 7 and 8 months. Currently, axicabtagene ciloleucel, tisagenlecleucel and lisocabtagene maraleucel are approved in the third line for patients with R/R DLBCL.
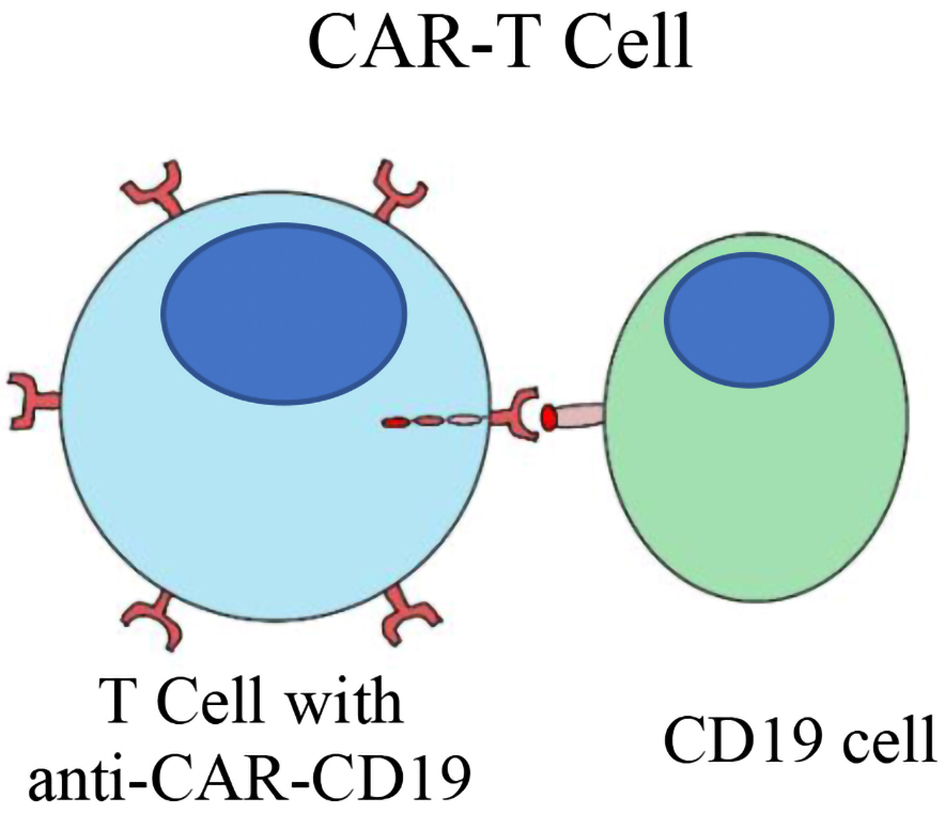 Click for large image | Figure 6. CARs and tumor cell interaction. CARs: chimeric antigen receptor T cells. |
Axicabtagene ciloleucel
Axicabtagene ciloleucel (axi-cel), an autologous anti-CD19 CARs therapy, showed efficacy in patients with R/R DLBCL. In an international phase III trial, 180 patients with large B-cell lymphoma were randomly assigned to receive axi-cel or standard care. A response occurred in 83% of patients receiving axi-cel and 50% of patients receiving standard care, with a CR in 65% of patients receiving axi-cel. Patients were randomly assigned to receive axi-cel or standard care (two or three cycles of investigator-selected, protocol-defined chemoimmunotherapy, followed by high-dose chemotherapy with ASCT in patients with a response to the chemoimmunotherapy). The median EFS was 8.3 months in the axi-cel group and 2.0 months in the standard-care group, and the 24-month EFS was 41% and 16%, respectively. In an interim analysis, the estimated OS at 2 years was 61% in the axi-cel group and 52% in the standard-care group. No deaths related to cytokine release syndrome or neurologic events occurred [100].
Tisagenlecleucel
Tisagenlecleucel (tisa-cel) was the first approved CARs therapy for childhood and adult relapsed or refractory B-ALL and later for adult R/R DLBCL. In an international phase III trial, 95.7% of 322 patients who received tisa-cel had a response, compared to 32.5% of patients who received salvage chemotherapy and autologous hematopoietic stem-cell transplantation. Ten patients in the tisa-cel group and 13 in the standard-care group died from adverse events. The median EFS in both groups was 3.0 months (HR for event or death in the tisa-cel group, 1.07; 95% CI: 0.82 - 1.40; P = 0.61). It was not superior to standard salvage therapy in this phase III trial [7].
Lisocabtagene maraleucel
Lisocabtagene maraleucel (liso-cel) is an autologous, CD19-directed, chimeric antigen receptor product that was studied in patients with R/R large B-cell lymphoma. It is an effective second-line treatment for patients with R/R DLBCL. In a global, phase III study with liso-cel versus SOC with salvage chemotherapy followed by ASCT as second-line treatment in patients with R/R DLBCL 232 patients were screened and 184 were assigned to the liso-cel (n = 92) or SOC (n = 92) groups. The median EFS was significantly improved in the liso-cel group (10.1 months, 95% CI: 6.1 - NR) compared with the SOC group (2.3 months) with P < 0.0001. There were no treatment-related deaths in the liso-cel group and one treatment-related death due to sepsis in the SOC group. The authors concluded liso-cel as an effective new second-line treatment in patients with early R/R DLBCL [8].
In an indirect comparison of tisa-cel and liso-cel R/R DLBCL the OS, PFS, CRR, and ORR were similar. The estimated adjusted 2-year OS, 2-year PFS, ORR, and CRR were 45.6%, 38.2%, 62.9%, and 47.7%, respectively, for tisa-cel versus 43.8%, 42.1%, 72.7%, and 53.1% for liso-cel. No significant differences in CR rate were observed [101]. The clinical outcome of 25% in R/R DLBCL who achieved remission at 3 and 6 months with CARs (73.8% and 86.5%, respectively) was maintained on long-term follow-up at 40.3-month period [102].
As second-line therapy for transplant-eligible R/R DLBCL within 12 months of completing chemo-immunotherapy with SOC, phase III clinical trials comparing axi-cel, tisa-cel, and liso-cel may change the treatment algorithm for DLBCL. A total of 437 patients were screened and 359 were randomized to axi-cel and 179 to SOC in the axi-cel (ZUMA-7) study. With a median follow-up of 24.9 months, the ZUMA-7 trial met its primary end point with a median EFS of 8.3 months (95% CI: 4.5 - 15.8) for axi-cel and 2 months (95% CI: 1.6 - 2.8) for SOC, with an HR of 0.40 (95% CI: 0.31 - 0.51; P < 0.001) [100]. There were 322 patients in the tisa-cel versus SOC (BELINDA) trial, with 162 randomized to tisa-cel and 160 randomized to SOC. After a median follow-up of 10 months, the BELINDA trial failed to meet its primary end point, with a median EFS of 3 months in both groups (HR, 1.07; 95% CI: 0.82 - 1.4; P = 0.61) [7]. In the liso-cel versus SOC (TRANSFORM) trial, 92 received liso-cel and 92 received SOC. After a median follow-up of 6.2 months, the TRANSFORM trial met its primary end point, with a median EFS of 10.1 months for liso-cel and 2.3 months for SOC (HR, 0.35) and a 65% risk reduction (P < 0.001) [8].
For patients who are ineligible for CARs therapy because of the urgent need for treatment (the median time between leukapheresis and delivery for axi-cel was 17 days and for tisa-cel 54 days) or failure to collect enough T cells to manufacture of CARs, bispecific antibodies may be an option, as these are readily available. Following bispecific antibodies, T cells can be collected effectively, CARs can be synthesized and expanded for successful CARs therapy [103].
Safety and efficacy of CARs therapy and checkpoint inhibitor therapy
A combination therapy with CD19 CARs and an anti-PD-1 antibody nivolumab was evaluated in patients with R/R DLBCL. The ORR and CR rate were 81.81% (9/11) and 45.45% (5/11), respectively. The median follow-up time was 6 months [104]. In the case of a PMBCL who experienced relapse 3.5 months after axi-cel, the patient received pembrolizumab (after four cycles) and complete metabolic response was confirmed [105]. In a study, 14 patients with heavily pretreated, relapsed B-ALL or B lymphoblastic lymphoma were treated with CD19-directed CARs in combination with pembrolizumab or nivolumab. In this study promising responses were seen in those with early B-cell recovery and bulky extramedullary disease. In contrast, PD-1 inhibition had partial, but no durable, effect in the four B-ALL patients with poor initial marrow response to CARs alone, suggesting a different mechanism may be responsible for poor initial responses [106]. In a study of 12 patients who received CART19 for R/R DLBCL enrolled with progressive disease (n = 8) or relapse (n = 4) following CART19, 11 patients had DLBCL (four germinal center (three “double/triple hit”), four non-germinal center, one T-cell rich DLBCL, one transformed FL, one primary mediastinal B-cell lymphoma) and one patient with FL were sequentially given CARs followed by pembrolizumab. The best ORR was 27% after pembrolizumab, with one patient having a CR, two PRs, and seven patients having progressive disease [107]. In a study assessing the efficacy and toxicity of CAR-T in combination with durvalumab, 12 of 13 patients with R/R DLBCL who received the combination therapy were evaluable for response. The ORR was 50% (five CR, 42%; one PR, 8%). Of the five patients who achieved CR, three patients were in CR at the first restaging after JCAR014 and two patients subsequently converted to CR after the first post-JCAR014 durvalumab infusion. Only one patient who achieved CR has relapsed (median follow-up of 10.6 months, range 3.7 - 11.8). Continued stable disease or evidence of regression was seen in four of six (67%) [108].
Chimeric antigen receptor failure
Immune escape is the primary mechanism from loss or downregulation of the target antigen. If CD19 CARs fail because of loss of CD19, the immune reconstitution can be restored by CD22 CARs [109]. However, if CARs still express CD19, then the immune response can be restored by taking advantage of checkpoint inhibitor therapy from PD1-PDL1 pathway interaction [110]. It has been documented that the checkpoint proteins PD1 and PD-L1 expressed on CARs and in the tumor microenvironment is up-regulated after infusion of the CAR product [111].
| Conclusion | ▴Top |
Beneficial immunological effect of “graft-versus-lymphoma” following allogeneic bone marrow transplantation is well established and led to other immunological interventions in DLBCL. The graft-versus-lymphoma was later taken advantage of by the “donor lymphocyte infusion”, albeit with limited success. The immune system can now be manipulated in other ways to alter the course of DLBCL. In the last decade, with immune-based therapies we have made rapid advances in the management of DLBCL. Ever since the advent of rituximab, incremental increases have been made in the management of DLBCL, leading to improved OS. We need standard guidelines to optimize and best utilize these agents in the management of R/R DLBCL
Acknowledgments
We acknowledge the help of Matt Torrence, the librarian of the University of South Florida for help with the literature search. We also acknowledge Ms. Preetha Chandreshwar for the illustrations in this manuscript
Financial Disclosure
No specific funding was received from public, commercial, or not-for-profit sectors to carry out the work described in this article.
Conflict of Interest
NV has been on the advisory board of Biogen Idec. Other authors declare there is no conflict of interest in writing this manuscript.
Author Contributions
NV did the literature search and RK wrote the initial version of the article. JR reviewed the article and AM revised the last version of the manuscript. All the authors approved the manuscript for submission.
Data Availability
The authors declare that the data supporting the findings of this study are available within the article.
| References | ▴Top |
- Mounier N, Heutte N, Thieblemont C, Briere J, Gaulard P, Feugier P, Ghesquieres H, et al. Ten-year relative survival and causes of death in elderly patients treated with R-CHOP or CHOP in the GELA LNH-985 trial. Clin Lymphoma Myeloma Leuk. 2012;12(3):151-154.
doi pubmed - Sehn LH, Martelli M, Trneny M, Liu W, Bolen CR, Knapp A, Sahin D, et al. A randomized, open-label, Phase III study of obinutuzumab or rituximab plus CHOP in patients with previously untreated diffuse large B-Cell lymphoma: final analysis of GOYA. J Hematol Oncol. 2020;13(1):71.
doi pubmed pmc - Salles G, Duell J, Gonzalez Barca E, Tournilhac O, Jurczak W, Liberati AM, Nagy Z, et al. Tafasitamab plus lenalidomide in relapsed or refractory diffuse large B-cell lymphoma (L-MIND): a multicentre, prospective, single-arm, phase 2 study. Lancet Oncol. 2020;21(7):978-988.
doi pubmed - Lurain K, Ramaswami R, Mangusan R, Widell A, Ekwede I, George J, Ambinder R, et al. Use of pembrolizumab with or without pomalidomide in HIV-associated non-Hodgkin's lymphoma. J Immunother Cancer. 2021;9(2):e002097.
doi pubmed pmc - Coiffier B, Thieblemont C, Van Den Neste E, Lepeu G, Plantier I, Castaigne S, Lefort S, et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: a study by the Groupe d'Etudes des Lymphomes de l'Adulte. Blood. 2010;116(12):2040-2045.
doi pubmed pmc - Cartron G, Hourcade-Potelleret F, Morschhauser F, Salles G, Wenger M, Truppel-Hartmann A, Carlile DJ. Rationale for optimal obinutuzumab/GA101 dosing regimen in B-cell non-Hodgkin lymphoma. Haematologica. 2016;101(2):226-234.
doi pubmed pmc - Bishop MR, Dickinson M, Purtill D, Barba P, Santoro A, Hamad N, Kato K, et al. Second-Line Tisagenlecleucel or Standard Care in Aggressive B-Cell Lymphoma. N Engl J Med. 2022;386(7):629-639.
doi pubmed - Kamdar M, Solomon SR, Arnason J, Johnston PB, Glass B, Bachanova V, Ibrahimi S, et al. Lisocabtagene maraleucel versus standard of care with salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment in patients with relapsed or refractory large B-cell lymphoma (TRANSFORM): results from an interim analysis of an open-label, randomised, phase 3 trial. Lancet. 2022;399(10343):2294-2308.
doi pubmed - Carlo-Stella C, Zinzani PLL, Janakiram M, Dia V, He X, Ervin-Haynes A, Depaus J. Planned interim analysis of a phase 2 study of loncastuximab tesirine plus ibrutinib in patients with advanced diffuse large B-cell lymphoma (LOTIS-3). Blood. 2021;138:54
- Jurczak W, Zinzani PL, Gaidano G, Goy A, Provencio M, Nagy Z, Robak T, et al. Phase IIa study of the CD19 antibody MOR208 in patients with relapsed or refractory B-cell non-Hodgkin's lymphoma. Ann Oncol. 2018;29(5):1266-1272.
doi pubmed pmc - Goebeler ME, Bargou R. Blinatumomab: a CD19/CD3 bispecific T cell engager (BiTE) with unique anti-tumor efficacy. Leuk Lymphoma. 2016;57(5):1021-1032.
doi pubmed - Coiffier B, Radford J, Bosly A, Martinelli G, Verhoef G, Barca G, Davies A, et al. A multicentre, phase II trial of ofatumumab monotherapy in relapsed/progressive diffuse large B-cell lymphoma. Br J Haematol. 2013;163(3):334-342.
doi pubmed - Schuster SJ, Bartlett NL, Assouline S, Yoon S-S, Bosch F, Sehn LH, Cheah CY, et al. Mosunetuzumab induces complete remissions in poor prognosis non-Hodgkin lymphoma patients, including those who are resistant to or relapsing after chimeric antigen receptor T-cell (CAR-T) therapies, and is active in treatment through multiple lines. Blood. 2019;134:6.
- Dickinson M, Carlo-Stella C, Morschhauser F, Bachy E, Corradini P, Iacoboni G, Khan C, et al. Glofitamab in patients with relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL) and ≥ 2 prior therapies: pivotal phase II expansion results. J Clin Oncol. 2022;40:7500.
- Michot J-M, Koh Y, Lee P, Jin J, Clynes R, Kye S, Chiarella M, et al. PB2102: phase 2 randomized, open-label, multicenter study to evaluate the efficacy and safety of plamotamab combined with tafasitamab (TAFA) + lenalidomide (LEN) VS TAFA + LEN in relapsed or refractory DLBCL. HemaSphere. 2022;6:1973-1974.
- Hutchings M, Mous R, Clausen MR, Johnson P, Linton KM, Chamuleau MED, Lewis DJ, et al. Dose escalation of subcutaneous epcoritamab in patients with relapsed or refractory B-cell non-Hodgkin lymphoma: an open-label, phase 1/2 study. Lancet. 2021;398(10306):1157-1169.
doi pubmed - Bannerji R, Arnason JE, Advani RH, Brown JR, Allan JN, Ansell SM, Barnes JA, et al. Odronextamab, a human CD20xCD3 bispecific antibody in patients with CD20-positive B-cell malignancies (ELM-1): results from the relapsed or refractory non-Hodgkin lymphoma cohort in a single-arm, multicentre, phase 1 trial. Lancet Haematol. 2022;9(5):e327-e339.
doi pubmed - Lamb YN. Inotuzumab Ozogamicin: First Global Approval. Drugs. 2017;77(14):1603-1610.
doi pubmed - Jacobsen ED, Sharman JP, Oki Y, Advani RH, Winter JN, Bello CM, Spitzer G, et al. Brentuximab vedotin demonstrates objective responses in a phase 2 study of relapsed/refractory DLBCL with variable CD30 expression. Blood. 2015;125(9):1394-1402.
doi pubmed - Northend M, Wilson W, Osborne W, Fox C, Davies A, El-Sharkawi D, Phillips E, et al. Polatuzumab vedotin with bendamustine and rituximab for relapsed/refractory high-grade B-cell lymphoma: the UK experience. Hematological Oncology. 2021;39.
- Advani R, Flinn I, Popplewell L, Forero A, Bartlett NL, Ghosh N, Kline J, et al. CD47 Blockade by Hu5F9-G4 and Rituximab in Non-Hodgkin's Lymphoma. N Engl J Med. 2018;379(18):1711-1721.
doi pubmed pmc - Zinzani PL, Thieblemont C, Melnichenko V, Bouabdallah K, Walewski J, Majlis A, Fogliatto L, et al. Efficacy and safety of pembrolizumab in relapsed/refractory primary mediastinal large B-cell lymphoma (rrPMBCL): updated analysis of the keynote-170 phase 2 trial. Blood. 2017;130:2833.
- Lesokhin AM, Ansell SM, Armand P, Scott EC, Halwani A, Gutierrez M, Millenson MM, et al. Nivolumab in Patients With Relapsed or Refractory Hematologic Malignancy: Preliminary Results of a Phase Ib Study. J Clin Oncol. 2016;34(23):2698-2704.
doi pubmed pmc - Hawkes EA, Phillips T, Budde LE, Santoro A, Saba NS, Roncolato F, Gregory GP, et al. Avelumab in Combination Regimens for Relapsed/Refractory DLBCL: Results from the Phase Ib JAVELIN DLBCL Study. Target Oncol. 2021;16(6):761-771.
doi pubmed pmc - Ribrag V, Lee ST, Rizzieri D, Dyer MJS, Fayad L, Kurzrock R, Andritsos L, et al. A Phase 1b Study to Evaluate the Safety and Efficacy of Durvalumab in Combination With Tremelimumab or Danvatirsen in Patients With Relapsed or Refractory Diffuse Large B-Cell Lymphoma. Clin Lymphoma Myeloma Leuk. 2021;21(5):309-317.e303.
doi pubmed - Solomon LA, Batista CR, DeKoter RP. Lenalidomide modulates gene expression in human ABC-DLBCL cells by regulating IKAROS interaction with an intronic control region of SPIB. Exp Hematol. 2017;56:46-57.e41.
doi pubmed - Gribben JG, Fowler N, Morschhauser F. Mechanisms of Action of Lenalidomide in B-Cell Non-Hodgkin Lymphoma. J Clin Oncol. 2015;33(25):2803-2811.
doi pubmed pmc - Nowakowski GS, Chiappella A, Witzig TE, Spina M, Gascoyne RD, Zhang L, Flament J, et al. ROBUST: Lenalidomide-R-CHOP versus placebo-R-CHOP in previously untreated ABC-type diffuse large B-cell lymphoma. Future Oncol. 2016;12(13):1553-1563.
doi pubmed pmc - Goy A, Ramchandren R, Ghosh N, Munoz J, Morgan DS, Dang NH, Knapp M, et al. Ibrutinib plus lenalidomide and rituximab has promising activity in relapsed/refractory non-germinal center B-cell-like DLBCL. Blood. 2019;134(13):1024-1036.
doi pubmed pmc - Westin J, Davis RE, Feng L, Hagemeister F, Steiner R, Lee HJ, Fayad L, et al. Smart Start: Rituximab, Lenalidomide, and Ibrutinib in Patients With Newly Diagnosed Large B-Cell Lymphoma. J Clin Oncol. 2023;41(4):745-755.
doi pubmed - Grommes C, Piotrowski A, Pentsova E, Nolan C, Francis J, DeAngelis L, Schaff L, et al. Phase Ib trial with dose expansion of the Bruton's Tyrosine Kinase (BTK) inhibitor, ibrutinib, in combination with rituximab and lenalidomide in patients with refractory/recurrent primary central nervous system lymphoma (PCNSL) and refractory/recurrent secondary central nervous system lymphoma (SCNSL). Blood. 2020l136:48.
- Geyer MB, Shaffer BC, Bhatnagar B, Mims AS, Klein V, Dilip D, Glass JL, et al. Lenalidomide-associated B-cell ALL: clinical and pathologic correlates and sensitivity to lenalidomide withdrawal. Blood Adv. 2023.
doi pubmed - Mato AR, Schuster SJ, Foss FM, Isufi I, Ding W, Brander DM, Sitlinger A, et al. A phase Ia/Ib study exploring the synthetic lethality of the orally administered novel BTK inhibitor, Dtrmwxhs-12 (DTRM-12), in combination with everolimus and pomalidomide in patients with relapsed/refractory CLL, DLBCL or other B-cell lymphomas. Blood. 2019;134:810.
- Reff ME, Carner K, Chambers KS, Chinn PC, Leonard JE, Raab R, Newman RA, et al. Depletion of B cells in vivo by a chimeric mouse human monoclonal antibody to CD20. Blood. 1994;83(2):435-445.
pubmed - Vose JM, Link BK, Grossbard ML, Czuczman M, Grillo-Lopez A, Gilman P, Lowe A, et al. Phase II study of rituximab in combination with chop chemotherapy in patients with previously untreated, aggressive non-Hodgkin's lymphoma. J Clin Oncol. 2001;19(2):389-397.
doi pubmed - Sehn LH, Donaldson J, Chhanabhai M, Fitzgerald C, Gill K, Klasa R, MacPherson N, et al. Introduction of combined CHOP plus rituximab therapy dramatically improved outcome of diffuse large B-cell lymphoma in British Columbia. J Clin Oncol. 2005;23(22):5027-5033.
doi pubmed - Pfreundschuh M, Schubert J, Ziepert M, Schmits R, Mohren M, Lengfelder E, Reiser M, et al. Six versus eight cycles of bi-weekly CHOP-14 with or without rituximab in elderly patients with aggressive CD20+ B-cell lymphomas: a randomised controlled trial (RICOVER-60). Lancet Oncol. 2008;9(2):105-116.
doi pubmed - Witzens-Harig M, Benner A, McClanahan F, Klemmer J, Brandt J, Brants E, Rieger M, et al. Rituximab maintenance improves survival in male patients with diffuse large B-cell lymphoma. Results of the HD2002 prospective multicentre randomized phase III trial. Br J Haematol. 2015;171(5):710-719.
doi pubmed - Morschhauser FA, Cartron G, Thieblemont C, Solal-Celigny P, Haioun C, Bouabdallah R, Feugier P, et al. Obinutuzumab (GA101) monotherapy in relapsed/refractory diffuse large b-cell lymphoma or mantle-cell lymphoma: results from the phase II GAUGUIN study. J Clin Oncol. 2013;31(23):2912-2919.
doi pubmed - Younes A, Burke JM, Cheson BD, Diefenbach C, Ferrari S, Hahn UH, Hawkes EA, et al. Safety and efficacy of atezolizumab in combination with rituximab plus CHOP in previously untreated patients with diffuse large B-cell lymphoma (DLBCL): updated analysis of a phase I/II study. Blood. 2019;134:2874.
- Vitolo U, Trneny M, Belada D, Carella AM, Chua N, Abrisqueta P, Demeter J, et al. Obinutuzumab or rituximab plus CHOP in patients with previously untreated diffuse large B-cell lymphoma: final results from an open-label, randomized phase 3 study (GOYA). Blood. 2016;128:470.
- Flinn IW, Erter J, Daniel DB, Mace JR, Berdeja JG. Phase II Study of Bendamustine and Ofatumumab in Elderly Patients with Newly Diagnosed Diffuse Large B-Cell Lymphoma Who Are Poor Candidates for R-CHOP Chemotherapy. Oncologist. 2019;24(8):1035-e1623.
doi pubmed pmc - Soe ZN, Allsup D. The use of ofatumumab in the treatment of B-cell malignancies. Future Oncol. 2017;13(29):2611-2628.
doi pubmed - Anderson KC, Bates MP, Slaughenhoupt BL, Pinkus GS, Schlossman SF, Nadler LM. Expression of human B cell-associated antigens on leukemias and lymphomas: a model of human B cell differentiation. Blood. 1984;63(6):1424-1433.
pubmed - Jaiswal S, Jamieson CH, Pang WW, Park CY, Chao MP, Majeti R, Traver D, et al. CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell. 2009;138(2):271-285.
doi pubmed pmc - Majeti R, Chao MP, Alizadeh AA, Pang WW, Jaiswal S, Gibbs KD, Jr., van Rooijen N, et al. CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell. 2009;138(2):286-299.
doi pubmed pmc - Zhang M, Hutter G, Kahn SA, Azad TD, Gholamin S, Xu CY, Liu J, et al. Anti-CD47 Treatment Stimulates Phagocytosis of Glioblastoma by M1 and M2 Polarized Macrophages and Promotes M1 Polarized Macrophages In Vivo. PLoS One. 2016;11(4):e0153550.
doi pubmed pmc - Muller D, Kontermann RE. Bispecific antibodies for cancer immunotherapy: Current perspectives. BioDrugs. 2010;24(2):89-98.
doi pubmed - Kontermann RE. Recombinant bispecific antibodies for cancer therapy. Acta Pharmacol Sin. 2005;26(1):1-9.
doi pubmed - Jabbour EJ, Short NJ, Jain N, Jammal N, Jorgensen J, Wang S, Wang X, et al. Blinatumomab is associated with favorable outcomes in patients with B-cell lineage acute lymphoblastic leukemia and positive measurable residual disease at a threshold of 10(-4) and higher. Am J Hematol. 2022;97(9):1135-1141.
doi pubmed - Katz DA, Morris JD, Chu MP, David KA, Thieblemont C, Morley NJ, Khan SS, et al. Open-label, phase 2 study of blinatumomab after frontline R-chemotherapy in adults with newly diagnosed, high-risk DLBCL. Leuk Lymphoma. 2022;63(9):2063-2073.
doi pubmed - Thompson PA, Jiang X, Banerjee P, Basar R, Garg N, Chen K, Kaplan M, et al. A phase two study of high dose blinatumomab in Richter's syndrome. Leukemia. 2022;36(9):2228-2232.
doi pubmed pmc - Olszewski AJ, Avigdor A, Babu S, Levi I, Abadi U, Holmes H, McKinney M, et al. Single-agent mosunetuzumab is a promising safe and efficacious chemotherapy-free regimen for elderly/unfit patients with previously untreated diffuse large B-cell lymphoma. Blood. 2020;136:43-45.
- Phillips TJ, Olszewski AJ, Munoz J, Kim TM, Yoon DH, Greil R, Westin J, et al. Mosunetuzumab, a novel CD20/CD3 bispecific antibody, in combination with CHOP confers high response rates in patients with diffuse large B-cell lymphoma. Blood. 2020;136:37-38.
- Patel K, Michot J-M, Chanan-Khan A, Ghesquieres H, Bouabdallah K, Byrd JC, Cartron G, et al. Safety and anti-tumor activity of plamotamab (XmAb13676), an anti-CD20 x anti-CD3 bispecific antibody, in subjects with relapsed/refractory non-Hodgkin's lymphoma. Blood. 2021;138:2494.
- van der Horst HJ, de Jonge AV, Hiemstra IH, Gelderloos AT, Berry DR, Hijmering NJ, van Essen HF, et al. Epcoritamab induces potent anti-tumor activity against malignant B-cells from patients with DLBCL, FL and MCL, irrespective of prior CD20 monoclonal antibody treatment. Blood cancer journal 2021, 11:1-8.
- Bannerji R, Allan JN, Arnason JE, Brown JR, Advani R, Ansell SM, O'Brien SM, et al. Odronextamab (REGN1979), a human CD20 x CD3 bispecific antibody, induces durable, complete responses in patients with highly refractory B-cell non-Hodgkin lymphoma, including patients refractory to CAR T therapy. Blood 2020, 136:42-43.
- Hutchings M, Morschhauser F, Iacoboni G, Carlo-Stella C, Offner FC, Sureda A, Salles G, et al. Glofitamab, a Novel, Bivalent CD20-Targeting T-Cell-Engaging Bispecific Antibody, Induces Durable Complete Remissions in Relapsed or Refractory B-Cell Lymphoma: A Phase I Trial. J Clin Oncol. 2021;39(18):1959-1970.
doi pubmed pmc - Sievers EL, Senter PD. Antibody-drug conjugates in cancer therapy. Annu Rev Med. 2013;64:15-29.
doi pubmed - Chu Y, Zhou X, Wang X. Antibody-drug conjugates for the treatment of lymphoma: clinical advances and latest progress. J Hematol Oncol. 2021;14(1):88.
doi pubmed pmc - Staudacher AH, Brown MP. Antibody drug conjugates and bystander killing: is antigen-dependent internalisation required? Br J Cancer. 2017;117(12):1736-1742.
doi pubmed pmc - Sloane BF, Yan S, Podgorski I, Linebaugh BE, Cher ML, Mai J, Cavallo-Medved D, et al. Cathepsin B and tumor proteolysis: contribution of the tumor microenvironment. Semin Cancer Biol. 2005;15(2):149-157.
doi pubmed - Reddy VY, Zhang QY, Weiss SJ. Pericellular mobilization of the tissue-destructive cysteine proteinases, cathepsins B, L, and S, by human monocyte-derived macrophages. Proc Natl Acad Sci U S A. 1995;92(9):3849-3853.
doi pubmed pmc - Kim SJ, Yoon DH, Kim JS, Kang HJ, Lee HW, Eom HS, Hong JY, et al. Efficacy of Brentuximab Vedotin in Relapsed or Refractory High-CD30-Expressing Non-Hodgkin Lymphomas: Results of a Multicenter, Open-Labeled Phase II Trial. Cancer Res Treat. 2020;52(2):374-387.
doi pubmed pmc - Zinzani PL, Pellegrini C, Chiappella A, Di Rocco A, Salvi F, Cabras MG, Argnani L, et al. Brentuximab vedotin in relapsed primary mediastinal large B-cell lymphoma: results from a phase 2 clinical trial. Blood. 2017;129(16):2328-2330.
doi pubmed - Bartlett NL, Smith MR, Siddiqi T, Advani RH, O'Connor OA, Sharman JP, Feldman T, et al. Brentuximab vedotin activity in diffuse large B-cell lymphoma with CD30 undetectable by visual assessment of conventional immunohistochemistry. Leuk Lymphoma. 2017;58(7):1607-1616.
doi pubmed - Bartlett NL, Yasenchak CA, Ashraf KK, Harwin WN, Sims RB, Nowakowski GS. Brentuximab vedotin in combination with lenalidomide and rituximab in subjects with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) (Trials in Progress). Wolters Kluwer Health. 2021.
- Bartlett NL, Yasenchak CA, Sims RB, Nowakowski GS. Trial-in-progress: brentuximab vedotin in combination with lenalidomide and rituximab in subjects with relapsed or refractory diffuse large B-cell lymphoma (DLBCL). Blood. 2020;136:14.
- Zinzani PL, Santoro A, Gritti G, Brice P, Barr PM, Kuruvilla J, Cunningham D, et al. Nivolumab Combined With Brentuximab Vedotin for Relapsed/Refractory Primary Mediastinal Large B-Cell Lymphoma: Efficacy and Safety From the Phase II CheckMate 436 Study. J Clin Oncol. 2019;37(33):3081-3089.
doi pubmed pmc - Ogura M, Tobinai K, Hatake K, Davies A, Crump M, Ananthakrishnan R, Ishibashi T, et al. Phase I Study of Inotuzumab Ozogamicin Combined with R-CVP for Relapsed/Refractory CD22+ B-cell Non-Hodgkin Lymphoma. Clin Cancer Res. 2016;22(19):4807-4816.
doi pubmed - Dang NH, Ogura M, Castaigne S, Fayad LE, Jerkeman M, Radford J, Pezzutto A, et al. Randomized, phase 3 trial of inotuzumab ozogamicin plus rituximab versus chemotherapy plus rituximab for relapsed/refractory aggressive B-cell non-Hodgkin lymphoma. Br J Haematol. 2018;182(4):583-586.
doi pubmed pmc - Sangha R, Davies A, Dang NH, Ogura M, MacDonald DA, Ananthakrishnan R, Paccagnella ML, et al. Phase 1 study of inotuzumab ozogamicin combined with R-GDP for the treatment of patients with relapsed/refractory CD22+ B-cell non-Hodgkin lymphoma. J Drug Assess. 2017;6(1):10-17.
doi pubmed pmc - Advani A, Coiffier B, Czuczman MS, Dreyling M, Foran J, Gine E, Gisselbrecht C, et al. Safety, pharmacokinetics, and preliminary clinical activity of inotuzumab ozogamicin, a novel immunoconjugate for the treatment of B-cell non-Hodgkin's lymphoma: results of a phase I study. J Clin Oncol. 2010;28(12):2085-2093.
doi pubmed - Wagner-Johnston ND, Goy A, Rodriguez MA, Ehmann WC, Hamlin PA, Radford J, Thieblemont C, et al. A phase 2 study of inotuzumab ozogamicin and rituximab, followed by autologous stem cell transplant in patients with relapsed/refractory diffuse large B-cell lymphoma. Leuk Lymphoma. 2015;56(10):2863-2869.
doi pubmed - Okazaki M, Luo Y, Han T, Yoshida M, Seon BK. Three new monoclonal antibodies that define a unique antigen associated with prolymphocytic leukemia/non-Hodgkin's lymphoma and are effectively internalized after binding to the cell surface antigen. Blood. 1993;81(1):84-94.
pubmed - Sehn LH, Hertzberg M, Opat S, Herrera AF, Assouline S, Flowers CR, Kim TM, et al. Polatuzumab vedotin plus bendamustine and rituximab in relapsed/refractory DLBCL: survival update and new extension cohort data. Blood Adv. 2022;6(2):533-543.
doi pubmed pmc - Herrera AF, Matasar MJ, Assouline S, Kamdar M, Mehta A, Fleury I, Kim WS, et al. Polatuzumab vedotin combined with bendamustine (B) and rituximab (R) or obinutuzumab (G) in patients with relapsed or refractory (R/R) follicular lymphoma (FL) or diffuse large B-cell lymphoma (DLBCL): preliminary results of a phase IB/II dose-escalation study. Blood. 2016;128:4194.
- Vodicka P, Benesova K, Janikova A, Prochazka V, Belada D, Mocikova H, Steinerova K, et al. Polatuzumab vedotin plus bendamustine and rituximab in patients with relapsed/refractory diffuse large B-cell lymphoma in the real world. Eur J Haematol. 2022;109(2):162-165.
doi pubmed - Segman Y, Ribakovsky E, Avigdor A, Goldhecht Y, Vainstein V, Goldschmidt N, Harlev S, et al. Outcome of relapsed/refractory diffuse large B-cell lymphoma patients treated with polatuzumab vedotin-based therapy: real-life experience. Leuk Lymphoma. 2021;62(1):118-124.
doi pubmed - Liebers N, Duell J, Fitzgerald D, Kerkhoff A, Noerenberg D, Kaebisch E, Acker F, et al. Polatuzumab vedotin as a salvage and bridging treatment in relapsed or refractory large B-cell lymphomas. Blood Adv. 2021;5(13):2707-2716.
doi pubmed pmc - Smith SD, Lopedote P, Samara Y, Mei M, Herrera AF, Winter AM, Hill BT, et al. Polatuzumab Vedotin for Relapsed/Refractory Aggressive B-cell Lymphoma: A Multicenter Post-marketing Analysis. Clin Lymphoma Myeloma Leuk. 2021;21(3):170-175.
doi pubmed - Gouni S, Rosenthal AC, Crombie JL, Ip A, Kamdar M, Hess BT, Feng L, et al. A multi-center retrospective study of polatuzumab for patients with large B-cell lymphoma relapsed after standard of care CAR T-cell therapy. Blood. 2021;138:2495.
- Lee A. Loncastuximab Tesirine: First Approval. Drugs. 2021;81(10):1229-1233.
doi pubmed - Hamadani M, Radford J, Carlo-Stella C, Caimi PF, Reid E, O'Connor OA, Feingold JM, et al. Final results of a phase 1 study of loncastuximab tesirine in relapsed/refractory B-cell non-Hodgkin lymphoma. Blood. 2021;137(19):2634-2645.
doi pubmed pmc - Hess B, Townsend W, Ai W, Stathis A, Solh M, Alderuccio JP, Ungar D, et al. Efficacy and Safety Exposure-Response Analysis of Loncastuximab Tesirine in Patients with B cell non-Hodgkin Lymphoma. AAPS J. 2021;24(1):11.
doi pubmed - Apostolidis J, Sayyed A, Darweesh M, Kaloyannidis P, Al Hashmi H. Current Clinical Applications and Future Perspectives of Immune Checkpoint Inhibitors in Non-Hodgkin Lymphoma. J Immunol Res. 2020;2020:9350272.
doi pubmed pmc - Merryman RW, Armand P, Wright KT, Rodig SJ. Checkpoint blockade in Hodgkin and non-Hodgkin lymphoma. Blood Adv. 2017;1(26):2643-2654.
doi pubmed pmc - Smith SD, Till BG, Shadman MS, Lynch RC, Cowan AJ, Wu QV, Voutsinas J, et al. Pembrolizumab with R-CHOP in previously untreated diffuse large B-cell lymphoma: potential for biomarker driven therapy. Br J Haematol. 2020;189(6):1119-1126.
doi pubmed - Chong EA, Melenhorst JJ, Svoboda J, Nasta SD, Landsburg DJ, Mato AR, Tian L, et al. Phase I/II study of pembrolizumab for progressive diffuse large B cell lymphoma after anti-CD19 directed chimeric antigen receptor modified T cell therapy. Blood. 2017;130:4121.
- Thornton PD, Bellas C, Santon A, Shah G, Pocock C, Wotherspoon AC, Matutes E, et al. Richter's transformation of chronic lymphocytic leukemia. The possible role of fludarabine and the Epstein-Barr virus in its pathogenesis. Leuk Res. 2005;29(4):389-395.
doi pubmed - Ding W, LaPlant BR, Call TG, Parikh SA, Leis JF, He R, Shanafelt TD, et al. Pembrolizumab in patients with CLL and Richter transformation or with relapsed CLL. Blood, The Journal of the American Society of Hematology. 2017;129:3419-3427.
- Ansell SM, Minnema MC, Johnson P, Timmerman JM, Armand P, Shipp MA, Rodig SJ, et al. Nivolumab for Relapsed/Refractory Diffuse Large B-Cell Lymphoma in Patients Ineligible for or Having Failed Autologous Transplantation: A Single-Arm, Phase II Study. J Clin Oncol. 2019;37(6):481-489.
doi pubmed pmc - Skarbnik AP, Donato ML, Korngold R, Feinman R, Rowley SD, Goy A, Vesole DH, et al. Safety and efficacy data for combined checkpoint inhibition with ipilimumab (Ipi) and nivolumab (Nivo) as consolidation following autologous stem cell transplantation (ASCT) for high-risk hematological malignancies-CPIT-001 trial. Blood. 2018;132:256.
- Hawkes EA, Chong G, Smith C, Lee S-T, Churilov L, McKendrick J, Renwick W, et al. Safety and efficacy of induction and maintenance avelumab plus R-CHOP in patients with diffuse large B-cell lymphoma (DLBCL): analysis of the Phase II Avr-CHOP Study. Blood. 2020;136:43-44.
- Nowakowski GS, Willenbacher W, Greil R, Larsen TS, Patel K, Jager U, Manges RF, et al. Safety and efficacy of durvalumab with R-CHOP or R(2)-CHOP in untreated, high-risk DLBCL: a phase 2, open-label trial. Int J Hematol. 2022;115(2):222-232.
doi pubmed - Vera JF, Brenner MK, Dotti G. Immunotherapy of human cancers using gene modified T lymphocytes. Curr Gene Ther. 2009;9(5):396-408.
doi pubmed pmc - Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR, Teachey DT, et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med. 2013;368(16):1509-1518.
doi pubmed pmc - Fitzgerald JC, Weiss SL, Maude SL, Barrett DM, Lacey SF, Melenhorst JJ, Shaw P, et al. Cytokine Release Syndrome After Chimeric Antigen Receptor T Cell Therapy for Acute Lymphoblastic Leukemia. Crit Care Med. 2017;45(2):e124-e131.
doi pubmed pmc - Brudno JN, Kochenderfer JN. Chimeric antigen receptor T-cell therapies for lymphoma. Nat Rev Clin Oncol. 2018;15(1):31-46.
doi pubmed - Locke FL, Miklos DB, Jacobson CA, Perales MA, Kersten MJ, Oluwole OO, Ghobadi A, et al. Axicabtagene Ciloleucel as Second-Line Therapy for Large B-Cell Lymphoma. N Engl J Med. 2022;386(7):640-654.
doi pubmed - Schuster SJ, Zhang J, Yang H, Agarwal A, Tang W, Martinez-Prieto M, Bollu V, et al. Comparative efficacy of tisagenlecleucel and lisocabtagene maraleucel among adults with relapsed/refractory large B-cell lymphomas: an indirect treatment comparison. Leuk Lymphoma. 2022;63(4):845-854.
doi pubmed - Ali A, Goy A, Dunleavy K. CAR T-cell therapy in highly aggressive B-cell lymphoma: emerging biological and clinical insights. Blood. 2022;140(13):1461-1469.
doi pubmed - Gonzalez Barca E. Role of Bispecific Antibodies in Relapsed/Refractory Diffuse Large B-Cell Lymphoma in the CART Era. Front Immunol. 2022;13:909008.
doi pubmed pmc - Cao Y, Lu W, Sun R, Jin X, Cheng L, He X, Wang L, et al. Anti-CD19 Chimeric Antigen Receptor T Cells in Combination With Nivolumab Are Safe and Effective Against Relapsed/Refractory B-Cell Non-hodgkin Lymphoma. Front Oncol. 2019;9:767.
doi pubmed pmc - Dimou M, Bitsani A, Bethge W, Panayiotidis P, Vassilakopoulos TP. Pembrolizumab-induced Remission After Failure of Axicabtagene Ciloleucel: Case Report and Literature Review. In Vivo. 2021;35(6):3401-3406.
doi pubmed pmc - Li AM, Hucks GE, Dinofia AM, Seif AE, Teachey DT, Baniewicz D, Callahan C, et al. Checkpoint inhibitors augment CD19-directed chimeric antigen receptor (CAR) T cell therapy in relapsed B-cell acute lymphoblastic leukemia. Blood. 2018;132:556.
- Chong EA, Svoboda J, Nasta SD, Landsburg DJ, Winchell N, Napier E, Mato AR, et al. Sequential anti-CD19 directed chimeric antigen receptor modified T-cell therapy (CART19) and PD-1 blockade with pembrolizumab in patients with relapsed or refractory B-cell non-Hodgkin lymphomas. Blood. 2018;132:4198.
- Hirayama AV, Gauthier J, Hay KA, Sheih A, Cherian S, Chen X, Pender BS, et al. Efficacy and toxicity of JCAR014 in combination with durvalumab for the treatment of patients with relapsed/refractory aggressive B-cell non-Hodgkin lymphoma. Blood. 2018;132:1680-1680.
- Fry TJ, Shah NN, Orentas RJ, Stetler-Stevenson M, Yuan CM, Ramakrishna S, Wolters P, et al. CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat Med. 2018;24(1):20-28.
doi pubmed pmc - Nie Y, Lu W, Chen D, Tu H, Guo Z, Zhou X, Li M, et al. Mechanisms underlying CD19-positive ALL relapse after anti-CD19 CAR T cell therapy and associated strategies. Biomark Res. 2020;8:18.
doi pubmed pmc - Cherkassky L, Morello A, Villena-Vargas J, Feng Y, Dimitrov DS, Jones DR, Sadelain M, et al. Human CAR T cells with cell-intrinsic PD-1 checkpoint blockade resist tumor-mediated inhibition. J Clin Invest. 2016;126(8):3130-3144.
doi pubmed pmc
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Hematology is published by Elmer Press Inc.


