| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website http://www.thejh.org |
Letter to the Editor
Volume 4, Number 2, June 2015, pages 184-186
Automated Detection and Classification of Schistocytes by a Novel Red Blood Cell Module Using Digital Imaging/Microscopy
Angelique Egelea, b, Warry van Geldera, Jurgen Riedla
aResult Laboratory, Albert Schweitzer Hospital, Dordrecht, The Netherlands
bCorresponding Author: Angelique Egele, Result Laboratory, Albert Schweitzer Hospital, Dordrecht, The Netherlands
Manuscript accepted for publication June 19, 2015
Short title: Detection of Schistocytes
doi: http://dx.doi.org/10.14740/jh214w
| To the Editor | ▴Top |
The detection of morphological red blood cell (RBC) abnormalities is of vital importance in a variety of diseases. For thrombotic thrombocytopenic purpura (TTP), the presence of schistocytes in a peripheral blood smear is a major criterion for the diagnosis. TTP is an acute life-threatening disease in which the protein ADAM-TS13 (a disintegrin and metalloprotease with thrombospondin type 1 motif, member 13) is ineffective (e.g. due to the presence of antibodies to ADAM-TS13). The function of this protein is cleavage of the coagulation protein von Willebrand factor (VWF) into smaller pieces [1]. Since ADAM-TS13 is inactive, blood clots are formed more rapidly which capture thrombocytes, resulting in a thrombocytopenia. Erythrocytes are fragmented by these clots, leading to hemolytic anemia and schistocytes. Patients often present with fever, hemorrhage, neurological symptoms, petechia and renal damage. Alongside, an increased lactate dehydrogenase (LDH) and a decreased haptoglobin are seen.
Schistocytes are morphologically polymorphic, which complicates the detection and identification. Also, a consistent and standardized international grading system for aberrant RBCs is still lacking nowadays [2]. The grading of abnormal RBCs is shown by a number of plus signs, which stands for the percentage of abnormal cells. In our laboratory, schistocytes are indicated with 1+, 2+ and 3+ (which stand for 0.5-2%, 2-5% and > 5% schistocytes) [3], but these percentages vary per laboratory. Recently a novel application module (Advanced RBC application, Cellavision) for the digital microscope was developed to automatically detect and classify morphological abnormalities of RBCs. This application actually works as a RBC-locating device. RBCs are identified and segmented by the system, after which each RBC is characterized for shape, size, color and inclusion. An artificial neural network is responsible for these characterizations, which is trained by a number of highly qualified experts. This neural network uses 80 features calculated for each RBC image. Some examples of used features are roundness, size, distribution of notches around the border and size and shape of inner pallor. The suggested morphology by the system can be changed or verified by the user. The results are represented as a percentage value and a grading which is based on a conversion table defined by the user in the settings file.
With the use of digital imaging and this novel RBC module, the morphological analysis of schistocytes could be significantly improved, resulting in a faster diagnosis.
Here we set out to evaluate and validate a novel RBC module capable of detection and classification of RBC abnormalities (e.g. schistocytes). For this, pre- and post-classification was performed on a cohort of patient samples and healthy controls. Pre-classification was performed by the RBC classification module without manual intervention (Fig. 1) and post-classification by a morphological expert. To determine correlation and accuracy, statistical analysis was conducted.
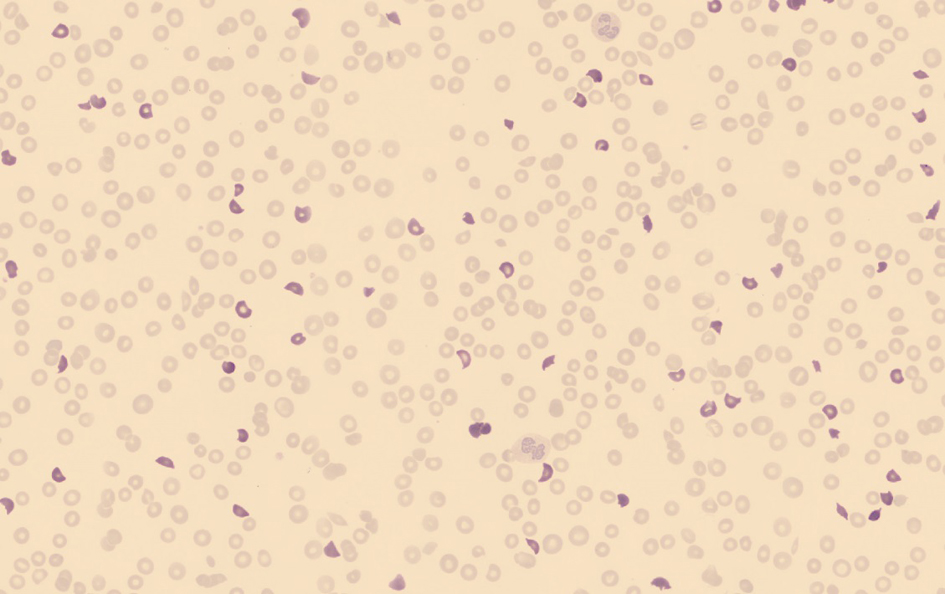 Click for large image | Figure 1. An overview of the red blood cells in a peripheral blood smear used for the classification in which the schistocytes (highlighted) are pre-classified by the software without manual intervention. |
Classification analysis of schistocytes was performed on 16 peripheral blood smears from patients in which schistocytes were present and a cohort of normal blood smears (n = 10). These samples were generated with the SP-10 (Slide-maker-stainer, Sysmex) from venous blood collected in EDTA tubes and stained according to the May-Grunwald Giemsa stain. Of these 16 patients, one was diagnosed with TTP. The remaining 15 samples included diagnoses such as thalassemia, solid tumors, myelofibrosis, myelodysplastic syndrome (MDS-RAEB 2), chronic lymphocytic leukemia (CLL), mantle cell lymphoma and some unknown (Table 1). Approximately 2,000 - 4,000 RBCs were classified per blood smear by the RBC module (Cellavision) in about 1 min. Statistical analysis was performed using the EP evaluator software from Siemens Healthcare Diagnostics and the Passing-Bablok method for regression analysis. The Passing-Bablok method is a linear regression procedure with no special assumptions regarding the distribution of the samples and the measurement errors [4].
 Click to view | Table 1. Pre-Classification and Post-Classification Results of the Schistocytes Classification in Patient Blood Smears (n = 16) |
Classification of the 16 samples resulted in a range of 1.2-19.0% schistocytes in the pre-classification and 0.5-12.3% schistocytes in the post-classification (Table 1). Pre- classification of the normal blood smears (n = 10) resulted in a range of 0.3-1.4% schistocytes and 0.1-0.2% in the post-classification. According to the VHL guidelines [3], eight of the 10 normal blood smears and none of the patient blood smears were false-positive, leading to a specificity of 80%. No false-negative samples were generated, leading to a sensitivity of 100%.
Statistical analysis showed a good correlation (Fig. 2) between the pre-classification and post-classification (correlation coefficient of 0.95 and y = 0.77x - 1.1), with approximately 20-25% over-estimation of schistocytes by the RBC module. Regression analysis showed a constant bias of -1.09% and a statistically significant proportional bias (1 - 0.773) with a confidence interval of 0.579 - 0.938. Reproducibility was tested using a sample with schistocytes (1.8%) and a sample without schistocytes. These samples were analyzed in 10 consecutive runs, resulting in a variation coefficient (VC) of 0.16% for the sample with schistocytes and a VC of 0.79% for the sample without schistocytes.
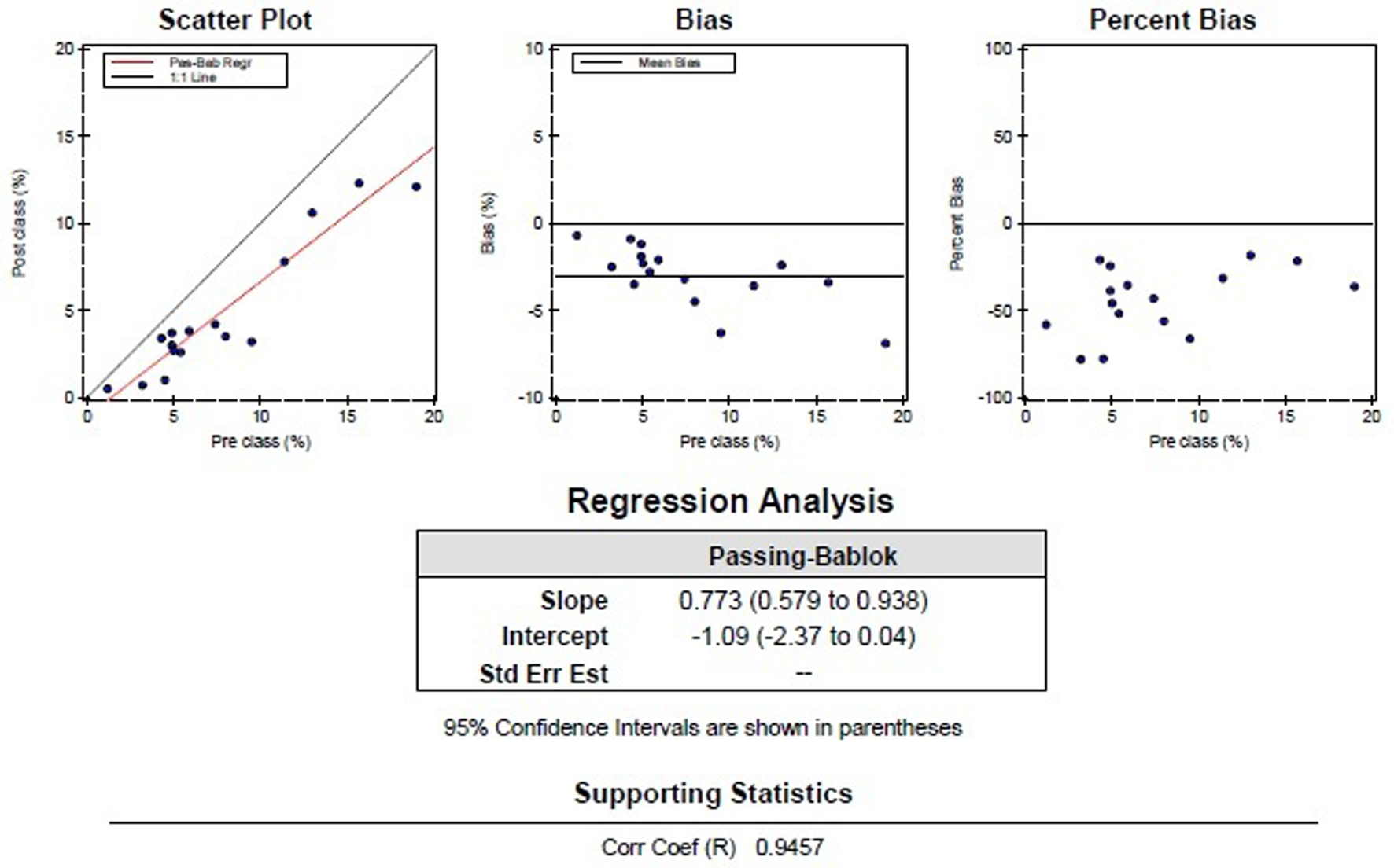 Click for large image | Figure 2. Graphic representation of the comparison of the pre-classification by the DM96 and post-classification by a morphological expert, with the normal samples excluded. |
Despite the polymorphic shape, recognition and classification of schistocytes by the RBC module is good. The pre- and post-classification of this aberrant morphological RBC shape also showed an excellent correlation (0.95). For healthy individuals, the pre-classification of schistocytes remained below 1.4%. For patients, the percentage of schistocytes started at 3.2%, with one exception of 1.2%. Amongst our patient samples a blood smear of a TTP-patient was present, which already showed 13.0% schistocytes in the pre-classification!
Automated morphological screening of blood smears using digital imaging in combination with cell-counter results (and schistocyte flagging) and a pre-classification limit set at approximately 1.0% schistocytes could help to distinguish diseases. More importantly, this could lead to a faster diagnosis of TTP, even without manual intervention.
| References | ▴Top |
- Sadler JE. Von Willebrand factor, ADAMTS13, and thrombotic thrombocytopenic purpura. Blood. 2008;112(1):11-18.
doi pubmed - Constantino BT. Reporting and grading of abnormal red blood cell morphology. Int J Lab Hematol. 2015;37(1):1-7.
doi pubmed - Ermens AAM, Mulder A, van Gelder W, van de Berg G. VHL. Diff magazine. 2013:15.
- Passing H, Bablok. A new biometrical procedure for testing the equality of measurements from two different analytical methods. Application of linear regression procedures for method comparison studies in clinical chemistry, Part I. J Clin Chem Clin Biochem. 1983;21(11):709-720.
pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Hematology is published by Elmer Press Inc.


