| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website http://www.thejh.org |
Case Report
Volume 4, Number 1, March 2015, pages 144-147
Enteropathy Type II Associated T-Cell Non-Hodgkin Lymphoma: A Case Report
Nisa Makruasia, d, Supap Makruasib, Therdkiat Trongwongsac
aDivision of Hematology, Department of Internal Medicine, Faculty of Medicine, Srinakharinwirot University, Nakhorn Nayok, Thailand
bSurgery Unit, Nakhorn Nayok Hospital, Thailand
cDepartment of Pathology, Faculty of Medicine, Srinakharinwirot University, Nakhorn Nayok, Thailand
dCorresponding Author: Nisa Makruasi, Division of Hematology, Department of Internal Medicine, Faculty of Medicine, Srinakharinwirot University, Nakhorn Nayok, Thailand
Manuscript accepted for publication February 17, 2015
Short title: T-cell Non-Hodgkin Lymphoma
doi: http://dx.doi.org/10.14740/jh186w
| Abstract | ▴Top |
Enteropathy-associated T-cell non-Hodgkin lymphoma (EATL) is a rare subtype of lymphoma, with aggressive course, unfavorable prognosis from resistance to chemotherapy and perforating. EATL type II is frequently found in Asia. We report the case that present with gastric perforation. Gastric involvement is rare. No first line standard treatment in this lymphoma is the cause of very rare subtype. The patient had good response after receiving chemotherapy but he died from septicemia.
Keywords: Non-Hodgkin lymphoma; T-cell lymphoma; Intestinal lymphoma
| Introduction | ▴Top |
Enteropathy-associated T-cell non-Hodgkin lymphoma (EATL) is uncommon, accounting for 5.4% of all non-Hodgkin lymphoma (NHL) [1]. It typically occurs in the small intestine, especially in the jejunum and ileum. The single or multiple ulcers in the small intestine, intestinal perforation or severe abdominal pain due to gut obstruction are common clinical features of EATL. Lymphoma may involve the spleen, liver, lymph nodes, stomach, gallbladder, colon or skin. The survival of EATL is dismal, since it behaves aggressively resistant to chemotherapy. This requires early diagnosis and high-intensity chemotherapy.
| Case Report | ▴Top |
A 60-year-old man presented with chronic epigastric pain, significant weight loss and night sweat for 4 months prior to visiting the hospital. On physical examination, he had mild pallor, no hepatosplenomegaly, mild tenderness at epigastrium, and no lymphadenopathy. A complete blood count revealed normal hematocrit, white cell count, and normal platelet count. Lactate dehydrogenase (LDH) level was 250 U/L. Examination with an esophagogastroduodenoscope showed multiple large ulcers in the body and antrum of stomach.
The tissue from the gastric mucosa showed dense and diffuse infiltration with small to medium sized immature lymphoid cells in the lamina propria which extended to submucosa; moreover, increased number of intraepithelial lymphocytes was also observed. No reactive lymphoid follicle was seen, as shown in Figure 1. These immature lymphoid cells were positive for CD3 and CD7 but negative for CD20, which indicated T cell in origin. The CD8 and TIA-1 stainings were positive, and CD56 positive lymphoma cells were detected, but weakly and focally positive. An aberrant phenotype (loss of expression of CD5) was also seen, as shown in Figure 2. Others stains including blast marker (TdT) and T-helper cell line (CD4) were negative as shown in Figure 2. The adjacent intraepithelial lymphocytes also had the same immunophenotype as the main lesion.
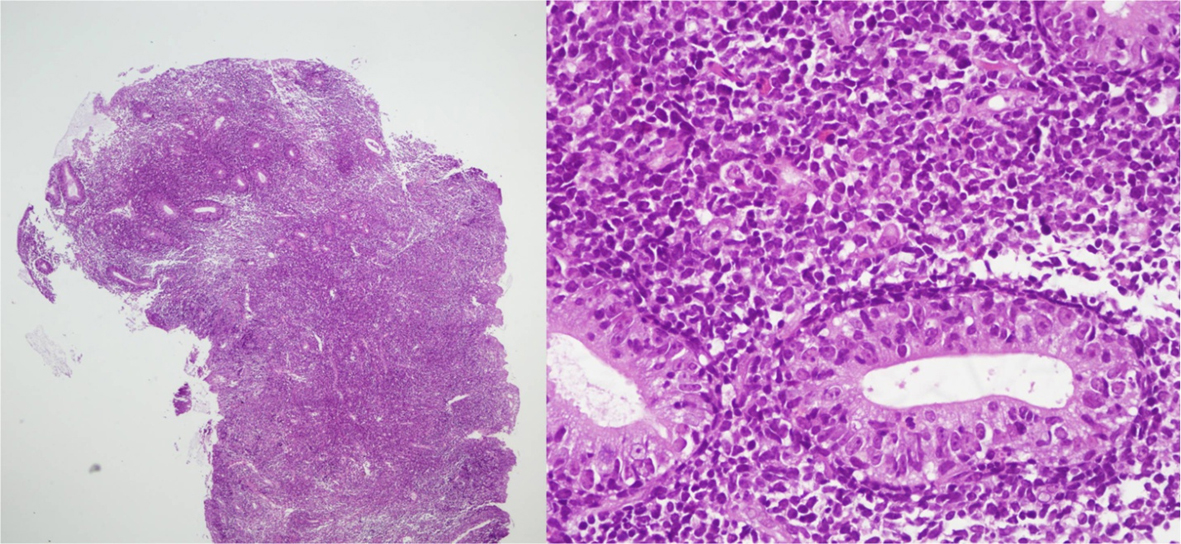 Click for large image | Figure 1. H&E slide shows abnormal small lymphocytes, infiltrated into mucosa and submucosa layer. The adjacent gastric glands show increase in intraepithelial lymphocytes. |
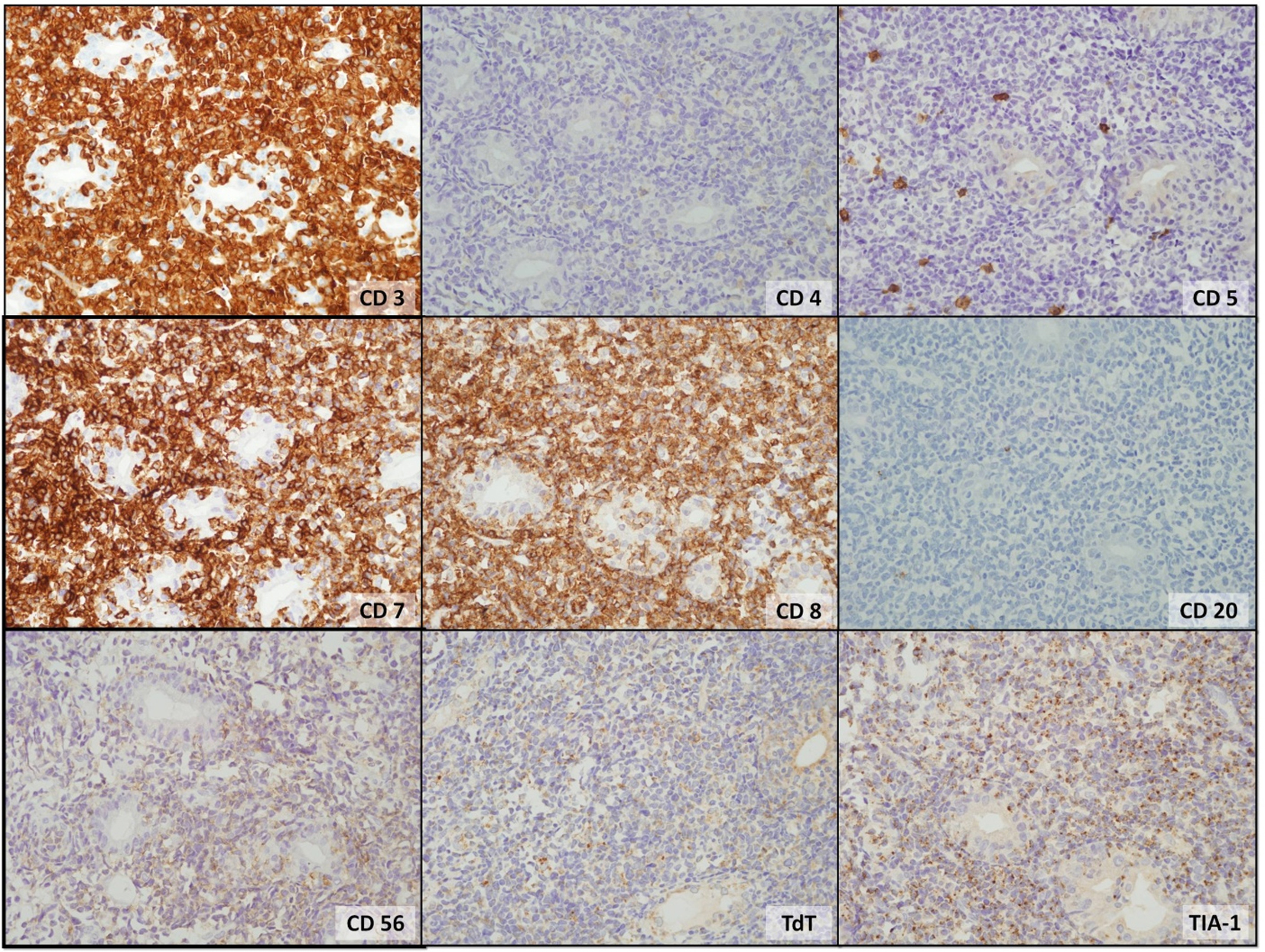 Click for large image | Figure 2. The tumor cells were strongly positive for CD3, CD7, CD8, and TIA-1. Weakly and focally positive for CD56, but negative for CD4, CD5, CD20 and TdT were detected. |
The evidences from the monotonous morphology, immunophenotype, and clinical history that this patient had no other primary lesions were fit for diagnostic criteria of EATL type II (WHO 2008), even the Epstein-Barr encoding region (EBER) in situ hybridization was negative.
Bone marrow pathology and immunohistochemistry illustrated interstitial and diffuse involvement by small to medium sized lymphoma cells.
Unfortunately, he developed sudden epigastric pain during waiting for the diagnosis. On physical examination, he had generalized peritonitis and the computerized tomography showed pneumoperitoneum, and multiple intra-abdominal lymphadenopathies. Exploratory laparotomy was performed, 2 cm in diameter of gastric perforation at cardia and jejunal perforation were seen intraoperatively. Subsequently repaired stomach with simple sutures, using an omental patch with biopsy and small bowel resection with end to end anastomosis was done.
Two weeks later, the endotracheal tube was taken off, and the surgical wound was healed already by physical examination. A complete blood count revealed hematocrit of 24%, low white cell count (1,500/µL with neutrophils 8.27%, lymphocytes 6.8%, monocytes 5.4%, eosinophils 0.2%, and basophils 0.4%) and low platelet count (68,000/µL). The LDH level was 788 U/L. CHOP regimen (cyclophosphamide, doxorubicin, vincristine, and prednisolone) was initiated. He developed febrile neutropenia with pseudomonas septicemia after the first course of chemotherapy; however, the cytopenia was improved and LDH levels returned to normal. He received three courses of the CHOP regimen with intrathecal methotrexate for CNS prophylaxis. Unfortunately, he died after the third cycles of CHOP from pseudomonas septicemia with unknown primary source of infection. The survival was 3 months after diagnosis.
| Discussion | ▴Top |
EATL is a rare subtype of NHL, with a high prevalence of coeliac disease associated with EATL type I, especially in northern Europe. EATL type I is more common than type II, occurring in 66% versus 34% of patients [1]. A monomorphic variant type (EATL type II) is frequently found in Asia while other types were rarely detected [2]. Approximately 90% of cases usually involve jejunum or ileum, but the duodenum involvement is rare [1]. Stomach and colon involvements are rare as well, accounting for only 8% and 16% of cases, respectively [1]. Bone marrow involvement is found in only 3% of cases [1]. The typical clinical presentation of EATL is abdominal pain which is often associated with intestinal perforation. The prognosis is very dismal due to chemotherapy resistance [3] and death from abdominal complications [4].
The tumor cells were positive for CD3, CD8, CD56, and TIA, but negative for CD 4, an immunophenotypic typical of type II EATL. A recent study of type II EATL showed about 8.8% EBV positive, the same as our case [5].
The peripheral T-cell lymphoma (PIT) prognostic index is a better survival predictor than the international prognostic index (IPI) [1]. The risk factors associated with poor overall survival are large tumor size (≥ 5 cm), non-ambulatory performance status leading to reduce chemotherapy intensity at the beginning of treatment, high levels of serum LDH and high C-reactive proteins reflecting extensive tissue damage, ulceration, perforation, and secondary inflammation [1]. The median overall survival (OS) is 10 months, and median failure-free survival (FFS) is 6 months [1]. The 5-year progression free survival (PFS) and the 5-year OS are 4% and 20%, respectively [6]. A standard treatment of EATL has not been developed due to the low incidence of this disease, differences in clinical presentation, and poor performance status. The role of surgery is only local debulking or resectioning the tumors showing a high risk of perforation, obstruction or hemorrhaging. These complications can occur also during chemotherapy or radiotherapy treatment [7], so postoperative complications should be carefully considered. The previous studies showed that patients who completed resectioning had a better prognosis than those who had residual disease [8-11]. Patients were treated with standard-dose multidrug chemotherapy. In about 35-40% of patients achieving complete remission [8, 9, 12, 13], their overall response rate ranges from 40% to 60%. The median duration of complete remission was 6 months [12, 14], with no difference between clinical stages. Only one study showed a median remission duration of 28 months (range: 17 - 39 months) and a median 2-year survival rate of 28% (range: 13-43%) [13]; however, the results were not statistically significant. The CHOP chemotherapy regimen was generally used and 5-year OS ranged from 9% to 22% [8, 12-15]. The OS and FFS were better in patients treated with anthracycline-based chemotherapy than other forms of treatment, including surgery or no treatment [1]. Other effective yet more toxic regimens include BACOP (bleomycin, doxorubicin, cyclophosphamide, vincristine, and prednisolone) [15], ProMACE-MOPP (prednisolone, doxorubicin, cyclophosphamide, etoposide, mechlorethamine, vincristine, and procarbazine) [15], VAMP (vincristine, doxorubicin, high-dose methotrexate, and prednisolone) [12], PEACE-BOM (prednisolone, etoposide, doxorubicin, cyclophosphamide-bleomycin, vincristine, and methotrexate) [12], and CHOEP (CHOP plus etoposide) [13]. Raderer et al reported six out of 19 refractory and relapsed patients after CHOP therapy were treated with ICE (two patients), DHAP (one patient) and fludarabine/cladribine and cyclophosphamide regimens (three patients). The complete remission was found in three patients (two from FC and one from ICE), two patients were still alive at 7 and 64 months, respectively, and another one died from disease progression after 10 months. The remaining three patients were not responded and died from perforation within 2 weeks after treatment with their second-line chemotherapy [11]. Treatment with ICE regimen followed by autologous stem cell transplantation showed significantly improved survival rates [14].
Based on Novakoic et al data, all patients treated with chemotherapy and radiotherapy regimen achieved complete remission [8]. Therefore, the combination of chemotherapy and radiotherapy should be considered for treatment of EATL. Another study involved novel therapy with alemtuzumab (monoclonal antibody against CD52), but the data were limited [16-18].
In conclusion, EATL is a rare disease with very poor outcomes. The best current treatment for patients who fit for chemotherapy is high-dose chemotherapy, preceded by surgical resection, and followed by ASCT.
Conflict of Interests
No financial or grant support or other assistance was received for this study.
| References | ▴Top |
- Delabie J, Holte H, Vose JM, Ullrich F, Jaffe ES, Savage KJ, Connors JM, et al. Enteropathy-associated T-cell lymphoma: clinical and histological findings from the international peripheral T-cell lymphoma project. Blood. 2011;118(1):148-155.
doi pubmed - Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. Lyon, France : IARC Press; 2008.
- Catassi C, Bearzi I, Holmes GK. Association of celiac disease and intestinal lymphomas and other cancers. Gastroenterology. 2005;128(4 Suppl 1):S79-86.
doi pubmed - van de Water JM, Cillessen SA, Visser OJ, Verbeek WH, Meijer CJ, Mulder CJ. Enteropathy associated T-cell lymphoma and its precursor lesions. Best Pract Res Clin Gastroenterol. 2010;24(1):43-56.
doi pubmed - Tse E, Gill H, Loong F, Kim SJ, Ng SB, Tang T, Ko YH, et al. Type II enteropathy-associated T-cell lymphoma: a multicenter analysis from the Asia Lymphoma Study Group. Am J Hematol. 2012;87(7):663-668.
doi pubmed - Savage KJ, Kahl BS. Non-Hodgkin lymphoma. In: McCrae KR, Steensma DP, eds.American Society of Hematology Self-Assessment Program 5th ed. Washington : American Society of Hematology; 2013;21:533-578.
- Di Sabatino A, Biagi F, Gobbi PG, Corazza GR. How I treat enteropathy-associated T-cell lymphoma. Blood. 2012;119(11):2458-2468.
doi pubmed - Novakovic BJ, Novakovic S, Frkovic-Grazio S. A single-center report on clinical features and treatment response in patients with intestinal T cell non-Hodgkin's lymphomas. Oncol Rep. 2006;16(1):191-195.
pubmed - Daum S, Ullrich R, Heise W, Dederke B, Foss HD, Stein H, Thiel E, et al. Intestinal non-Hodgkin's lymphoma: a multicenter prospective clinical study from the German Study Group on Intestinal non-Hodgkin's Lymphoma. J Clin Oncol. 2003;21(14):2740-2746.
doi pubmed - Honemann D, Prince HM, Hicks RJ, Seymour JF. Enteropathy-associated T-cell lymphoma without a prior diagnosis of coeliac disease: diagnostic dilemmas and management options. Ann Hematol. 2005;84(2):118-121.
doi pubmed - Raderer M, Troch M, Kiesewetter B, Puspok A, Jaeger U, Hoffmann M, Chott A. Second line chemotherapy in patients with enteropathy-associated T cell lymphoma: a retrospective single center analysis. Ann Hematol. 2012;91(1):57-61.
doi pubmed - Gale J, Simmonds PD, Mead GM, Sweetenham JW, Wright DH. Enteropathy-type intestinal T-cell lymphoma: clinical features and treatment of 31 patients in a single center. J Clin Oncol. 2000;18(4):795-803.
pubmed - Wohrer S, Chott A, Drach J, Puspok A, Hejna M, Hoffmann M, Raderer M. Chemotherapy with cyclophosphamide, doxorubicin, etoposide, vincristine and prednisone (CHOEP) is not effective in patients with enteropathy-type intestinal T-cell lymphoma. Ann Oncol. 2004;15(11):1680-1683.
doi pubmed - Sieniawski M, Angamuthu N, Boyd K, Chasty R, Davies J, Forsyth P, Jack F, et al. Evaluation of enteropathy-associated T-cell lymphoma comparing standard therapies with a novel regimen including autologous stem cell transplantation. Blood. 2010;115(18):3664-3670.
doi pubmed - Egan LJ, Walsh SV, Stevens FM, Connolly CE, Egan EL, McCarthy CF. Celiac-associated lymphoma. A single institution experience of 30 cases in the combination chemotherapy era. J Clin Gastroenterol. 1995;21(2):123-129.
doi pubmed - Gallamini A, Zaja F, Patti C, Billio A, Specchia MR, Tucci A, Levis A, et al. Alemtuzumab (Campath-1H) and CHOP chemotherapy as first-line treatment of peripheral T-cell lymphoma: results of a GITIL (Gruppo Italiano Terapie Innovative nei Linfomi) prospective multicenter trial. Blood. 2007;110(7):2316-2323.
doi pubmed - Soldini D, Mora O, Cavalli F, Zucca E, Mazzucchelli L. Efficacy of alemtuzumab and gemcitabine in a patient with enteropathy-type T-cell lymphoma. Br J Haematol. 2008;142(3):484-486.
doi pubmed - Kircher SM, Gurbuxani S, Smith SM. CHOP plus alemtuzumab can induce metabolic response by FDG-PET but has minimal long-term benefits: a case report and literature review. J Gastrointest Cancer. 2007;38(1):59-62.
doi pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Hematology is published by Elmer Press Inc.


